
.
21 May 2015
EMA/CAT/600280/2010 rev.1
Committee for Advanced Therapies (CAT)
Reflection paper on classification of advanced therapy
medicinal products
Draft Agreed by CAT June 2014
Adoption by CAT for release for consultation 20 June 2014
Start of public consultation 30 June 2014
End of consultation (deadline for comments) 31 October 2014
Draft Agreed by CAT 13 May 2015
Adoption by CAT 22 May 2015
Keywords
ATMP classification, Gene therapy, Somatic cell therapy, Tissue engineered
Products, Combined ATMPs
30 Churchill Place ● Canary Wharf ● London E14 5EU ● United Kingdom
An agency of the European Union
Telephone
+44 (0)20 3660 6000
Facsimile
+44 (0)20 3660 5555
Send a question via our website www.ema.europa.eu/contact
© European Medicines Agency, 2015. Reproduction is authorised provided the source is acknowledged.

Reflection paper on classification of Advanced Therapy
Medicinal Products
Table of contents
1. Executive summary ................................................................................. 3
2. Discussion ............................................................................................... 4
2.1. Legal basis of ATMP classification ........................................................................... 4
2.2. Scientific principles applied to the classification of ATMPs .......................................... 7
2.2.1. Definition of cell, viable cell and tissue for classification purposes ............................ 7
2.2.2. Claimed mode of action (MoA) ............................................................................ 7
2.2.3. Criteria for GTMP ............................................................................................... 8
2.2.4. Criteria for somatic cell therapy medicinal products (sCTMP) and tissue engineered
products (TEP) ......................................................................................................... 11
2.2.5. Criteria for combined ATMPs ............................................................................. 15
2.3. Evolving and borderlines areas ............................................................................. 15
2.3.1. Advanced therapies versus transplants/transfusion .............................................. 16
2.3.2. Classification of genetically modified bacteria as Gene therapy medicinal product .... 16
2.3.3. Gene therapy medicinal product versus cell therapy medicinal product ................... 17
2.3.4. Combined ATMPs versus non-combined ATMPs .................................................... 17
2.4. Clarifications on procedural aspects information to be submitted by the applicant ....... 18
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
2/19

1. Executive summary
Further to the implementation of Article 17 of Regulation (EC) No 1394/2007
i
(hereinafter referred as
to ‘the Advanced Therapy Medicinal Products (ATMPs) Regulation’), applicants have access to an
optional procedure which is the CAT (Committee for Advanced Therapies) scientific recommendation
for the classification of ATMPs, hereafter referred to as “ATMP classification”. It is underpinned by the
ATMP Regulation which enables the European Medicines Agency (EMA) in close collaboration with the
European Commission to determine whether or not a given product meets the scientific criteria, which
define ATMPs
ii.
The ATMP classification procedure has been established in order to address, as early as
possible, questions of borderline cases where classification of a product based on genes, cells or
tissues is not clear.
The CAT issues scientific recommendations determining whether or not the referred product falls,
within the definition of an ATMP in the European Union.
The ATMP Regulation and the Directive 2001/83/EC Annex I Part IV
ii
provide precise legal definitions
for ATMPs.
The ATMP classification is based on the evaluation of whether a given product fulfils one of the
definitions of gene therapy medicinal product (GTMP), somatic cell therapy medicinal product (sCTMP)
or tissue engineered product (TEP) and whether the product fulfils the definition of a combined ATMP
or not. However, it is also acknowledged that, due to the complex nature of these therapeutic products,
the limited data package at an early stage of product development and the rapid evolution of science
and technology, questions of borderline may arise
iii
.
The ATMP classification is conducted by the CAT on request of and on basis of information provided by
a developer of a product based on genes, cells or tissues and the outcome of the classification is
therefore specific to the product under development. The examples
1
and the conclusions mentioned in
this paper may not be directly applicable to other products which may be from a different origin or
manufactured using different processes / undergoing different manipulation steps. The CAT
classification examples in the reflection paper should not be understood as generic classifications for
certain classes of ATMPs. Future applicants should apply caution when extrapolating the CAT
classifications to their product and should consider applying for ATMP classification of their product.
The ATMP classification procedure is voluntary and free of charge. While the recommendation on
classification provided by the Agency is not binding, the procedure can help developers to clarify the
applicable regulatory framework. It also provides clarity on the development path and scientific-
regulatory guidance to be followed. The ATMP classification may sometimes also be a useful tool for
applicants to initiate a tailored dialogue on the product development with regulators. Indeed, the ATMP
classification, along with other tools (e.g. ITF briefing meetings
2
), should be seen as a first opportunity
to engage with regulators. Once the candidate ATMP classification has been clarified and confirmed,
the dialogue can continue with the use of other regulatory procedures such as scientific advice and
ATMP certification (only for small and medium enterprises). The ATMP classification may also help
developers to gain access to all relevant services and incentives offered by the EMA.
Although clinical trials are under the responsibility of the National Competent Authorities, it is
important to stress that the classification recommendation made by the CAT may help when submitting
a clinical trial dossier, as the applicant and the concerned competent authorities will be made aware of
1
In order to protect the commercially confidential information provided by the applicants, the examples in this reflection
paper are generalised, especially with regards to the manufacturing and manipulation steps.
2
See EMA website: European Medicines Agency - Human medicines - Innovation Task Force (ITF)
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
3/19

a European classification position which can clarify and facilitate identification of the most relevant
criteria and procedure to be applied.
Moreover, the ATMP classification can be applied for at any stage of the product development, even
when non-clinical and clinical data are not available. It should be noted that scientific
recommendations given by the CAT are always related to a defined product. It is thus not possible to
classify scientific ‘concepts’ in the absence of a clear description of the product.
In addition, the ATMP classification procedure is only applicable when a product is based on genes,
cells or tissues
3
.
If additional scientific information becomes available after the original ATMP classification that may
impact the classification of the product, the applicant can submit a follow-up request. This should
follow the same procedure as the original submission.
The summary outcome ATMP classifications assessed so far by the CAT is available on the EMA
website
4
. Since 2011, summary reports of all ATMP classifications are published.
Scope
The aim of this reflection paper is to provide guidance on the ATMP classification procedure, as well as
on the interpretation of key concepts of the definition of gene therapy medicinal product, somatic cell
therapy medicinal product, tissue engineered product, and combined advanced therapy medicinal
product. The guidance reflects the experience gained in the application of the classification procedure.
2. Discussion
2.1. Legal basis of ATMP classification
According to Article 2(1)(a) of Regulation (EC) No.1394/2007, an ‘advanced therapy medicinal product’
means any of the following medicinal products for human use:
• a gene therapy medicinal product as defined in Part IV of Annex I to Directive 2001/83/EC, as
amended
• a somatic cell therapy medicinal product as defined in Part IV of Annex I to Directive 2001/83/EC,
as amended
• a tissue engineered product as defined in Article 2(1)(b) of Regulation (EC) No. 1394/2007.
Article (2)(1)(d) of the ATMP Regulation also gives a definition of ‘Combined ATMP’. These products
contain as an integral part of the product a medical Device (see below).
The definitions of a gene therapy medicinal product and a somatic cell therapy medicinal product
according to Directive 2001/83/EC, Annex I, Part IV, as amended (implementing Directive
2009/120/EC) are as follows:
2.1.1. Gene therapy medicinal product
Gene therapy medicinal product means a biological medicinal product which has the following
characteristics:
3
Taking into account the remit of the European Medicines Agency, as stated in Article 17 of Regulation 1394/2007 i.e. “Any
applicant developing a product based on genes, cells or tissues may request a scientific recommendation of the Agency with
a view to determining whether the referred product falls, on scientific grounds, within the definition of an advanced therapy
medicinal product ….”
4
The complete list of scientific recommendations on classification of ATMPS can be found at:
http://www.ema.europa.eu/ema/index.jsp?curl=pages/regulation/general/general_content_000301.jsp&mid=WC0b01ac05
800862c0
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
4/19

(a) it contains an active substance which contains or consists of a recombinant nucleic acid
used in or administered to human beings with a view to regulating, repairing, replacing, adding
or deleting a genetic sequence;
(b) its therapeutic, prophylactic or diagnostic effect relates directly to the recombinant nucleic
acid sequence it contains, or to the product of genetic expression of this sequence.
Gene therapy medicinal products shall not include vaccines against infectious diseases.
It should be noted that in order to be considered a gene therapy medicinal product, both the
characteristics (a) and (b) have to be fulfilled.
2.1.2. Somatic cell therapy medicinal product
Somatic cell therapy medicinal product means a biological medicinal product which has the following
characteristics:
(a) contains or consists of cells or tissues that have been subject to substantial manipulation so
that biological characteristics, physiological functions or structural properties relevant for the
intended clinical use have been altered, or of cells or tissues that are not intended to be used
for the same essential function(s) in the recipient and the donor;
(b) is presented as having properties for, or is used in or administered to human beings with a
view to treating, preventing or diagnosing a disease through the pharmacological,
immunological or metabolic action of its cells or tissues.
For the purposes of point (a), the manipulations listed in Annex I to Regulation (EC) No 1394/2007, in
particular, shall not be considered as substantial manipulations: cutting, grinding, shaping,
centrifugation, soaking in antibiotic or antimicrobial solutions, sterilization, irradiation, cell separation,
concentration or purification, filtering, lyophilization, freezing, cryopreservation, and vitrification.
It should be pointed out that this list of non-substantial manipulations is non-exhaustive.
It should also be noted that in order to be considered a somatic cell therapy medicinal product, both
the characteristics (a) and (b) have to be fulfilled.
2.1.3. Tissue engineered products
Tissue engineered products, according to Article 2(1)(b) of Regulation (EC) No. 1394/2007, means a
product that:
- contains or consists of engineered cells or tissues, and
- is presented as having properties for, or is used in or administered to human beings
with a view to regenerating, repairing or replacing a human tissue.
A tissue engineered product may contain cells or tissues of human or animal origin, or
both. The cells or tissues may be viable or non-viable. It may also contain additional
substances, such as cellular products, bio-molecules, biomaterials, chemical substances,
scaffolds or matrices.
Products containing or consisting exclusively of non-viable human or animal cells and/or
tissues, which do not contain any viable cells or tissues and which do not act principally
by pharmacological, immunological or metabolic action, are excluded from this definition.
Article 2(1)(c) of Regulation (EC) No. 1394/2007 also states that:
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
5/19

Cells or tissues shall be considered ‘engineered’ if they fulfil at least one of the following
conditions:
- the cells or tissues have been subject to substantial manipulation, so that biological
characteristics, physiological functions or structural properties relevant for the intended
regeneration, repair or replacement are achieved. The manipulations listed in Annex I, in
particular, shall not be considered as substantial manipulations,
- the cells or tissues are not intended to be used for the same essential function or
functions in the recipient as in the donor.
2.1.4. Combined Advanced Therapy Medicinal Products
According to Article 2(1)(d) of Regulation (EC) No. 1394/2007, a ‘Combined advanced therapy
medicinal product’ means an advanced therapy medicinal product that fulfils the following conditions:
- it must incorporate, as an integral part of the product, one or more medical devices
within the meaning of Article 1(2)(a) of Directive 93/42/EEC or one or more active
implantable medical devices within the meaning of Article 1(2)(c) of Directive
90/385/EEC, and
- its cellular or tissue part must contain viable cells or tissues, or
- its cellular or tissue part containing non-viable cells or tissues must be liable to act
upon the human body with action that can be considered as primary to that of the
devices referred to.
For requirements for medical devices and implantable medical devices please consult the relevant
European Commission guidelines and Medical Device Legislation, as appropriate.
2.1.5 Additional legal clarifications in Regulation (EC) No. 1394/2007
• With regards to products containing cells or tissues, Article 2(2) states that:
“Where a product contains viable cells or tissues, the pharmacological, immunological or
metabolic action of those cells or tissues shall be considered as the principal mode of
action of the product.”
For Tissue Engineered products their Mode of Action is linked to regeneration, repair or
replacement a human tissue, as described in Article 2(1)(b).
• In accordance with Article 2(3), an advanced therapy medicinal product containing both
autologous and allogeneic cells or tissues shall be considered to be for allogeneic use.
• Demarcation rule between ATMPs:
Article 2(4) and 2(5) states that:
“A product which may fall within the definition of a tissue engineered product and within the
definition of a somatic cell therapy medicinal product shall be considered as a tissue
engineered product. A product which may fall within the definition of a somatic cell therapy
medicinal product or a tissue engineered product, and a gene therapy medicinal product, shall
be considered as a gene therapy medicinal product.”
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
6/19

2.2. Scientific principles applied to the classification of ATMPs
According to Article 17 of the ATMP Regulation, products are classified according to the respective
definitions of gene therapy medicinal product, somatic cell therapy medicinal products, tissue
engineered product and combined ATMP, on the basis of scientific information provided by the
applicant.
This section elucidates the scientific criteria applied for the classification of ATMPs. The following list of
criteria is based largely on the experience gained by the CAT through recommendations on ATMP
classification issued so far
4
. These should not be considered as exhaustive and might be subject to
change as science evolves.
2.2.1. Definition of cell, viable cell and tissue for classification purposes
For the purpose of ATMP classification, the CAT considers that a cell is defined as follows: ‘A typical cell
is the smallest unit of an organism that has been generated directly through mitosis. A cell comprises a
nucleus (eukaryotic cells) or nucleoid material (prokaryotic cells) and cytoplasma enclosed by a cell
membrane. A viable cell should be capable to produce energy and synthesize new molecules from raw
materials.’
This definition is to be read in conjunction with the relevant legislation including Regulation (EC) No
1394/2007 and Directive 2001/83/EC including its Annex I, part IV (technical requirements for ATMP).
A viable cell is a cell that has a functional cytoplasmic membrane. The European Pharmacopoeia
provides information on assays to demonstrate cytoplasmic membrane integrity and activity [Ph. Eur.
General Chapter 2.7.29 (01/2008:20729)]. In particular the concerned method refers to cell staining
by viability dyes and manual or automated analysis, under a light microscope or by flow cytometry, of
a cell suspension in order to determine the percentage of viable cells. The same definition of viable
cells is referred to in the ‘Guideline on human cell-based medicinal products’
(EMEA/CHMP/410869/2006)
Tissues are defined in Directive 2004/23/EC (Art 3.b) as ‘all constituent parts of a human body formed
by cells’.
2.2.2. Claimed mode of action (MoA)
Information on the claimed MoA is particularly important to ascertain whether the product is for
treatment, prevention or diagnosis of a disease, and exerts its activity via a pharmacological,
immunological or metabolic action, or whether the product is intended for regeneration, repair or
replacement of cells/tissues. The possible MoA should be considered in relation to the intended
indication.
For example, if mesenchymal stem cells are used to treat a diseased organ, this could act via a
combination of mechanisms which can include metabolic, immunological, pharmacological,
regeneration and repair. In such a case, the predominant mode of action claimed will affect whether
this will be classified as somatic cell therapy or tissue-engineered product.
The claim can be based either on data and/or on current scientific knowledge, but it has to be
sufficiently substantiated in each case. Otherwise, the CAT may only conclude that a product is an
ATMP, but not yet if it is, for example, a tissue engineered product or a somatic cell therapy medicinal
product.
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
7/19

2.2.3. Criteria for GTMP
The definition of gene therapy medicinal product according to Annex I, part IV, section 2.1 of Directive
2001/83/EC, as amended, is articulated into two conditions that have both to be fulfilled
simultaneously: 1) the product has to be a biological medicinal product
iv
and contains recombinant
nucleic acid(s) and 2) the recombinant nucleic acid(s) should be directly involved in the mechanism of
action (and hence therapeutic action of the product. In this respect the following observations can be
made:
• Indent (a) of the definition of Gene therapy medicinal product:
the recombinant nucleic acids should be of biological origin independently from the origin of the
vector system used (e.g. viral/bacterial vectors or micellar and liposomal formulations, etc.)
• Indent (b) of the definition of Gene therapy medicinal product :
“its therapeutic, prophylactic or diagnostic effect relates directly to the recombinant nucleic acid
sequence it contains, or to the product of genetic expression of this sequence”: the MoA and
proposed indication, as claimed by the applicant are of essential to assess if there is a “direct”
relationship between the therapeutic, prophylactic or diagnostic effect of the product and the
delivered genetic sequence or the expressed product. As an illustration, the CAT provided two
scientific recommendations for classifications for genetically modified T cells encoding an
exogenous thymidine kinase gene. The T cell preparations were intended for immune reconstitution
as adjunct treatment in haematopoietic stem cell transplantation.
These T cell preparations have been classified as somatic cell therapy medicinal products
considering that the treatment was adjunctive T-cell therapy supporting immune reconstitution of
leukaemia patients who underwent bone marrow transplantation after myeloablative conditioning
regime. In both cases, the genetic modification leading to the expression of the exogenous gene
herpes simplex virus thymidine kinase - by the addition of the corresponding genetic sequence -
relates to the treatment (with ganciclovir administration) of a potential graft versus host disease
that may occur in some patients undergoing Haematopoietic Stem Cell Therapy (HSCT). The
recommendation on the classification as somatic cell therapy considered that the primary role of
the cells was the “immune reconstitution” of the patients, while the genetic modification was
limited to a secondary role of controlling the potential risk of graft versus host disease. However, it
should be stressed that being considered as a genetically modified somatic cell therapy product,
most of the principles and requirements that normally apply to gene therapy medicinal products,
may also apply for these products (i.e. the classification does not necessarily exempt from the
relevant and applicable scientific requirements of GTMP).
• Genetic manipulation does not necessarily have to take place in the human body, since for
example products consisting of genetically modified cells generated ex-vivo have also been
classified as a gene therapy medicinal product (e.g. autologous CD34+ haematopoietic stem cells
(HSCs) transduced with lentiviral vector encoding the human ABCD1 cDNA and autologous CD34+
haematopoietic stem cells (HSCs) transduced with lentiviral vector encoding the human βA-T87Q-
globin gene).
• The legislation provides that “Gene therapy medicinal products shall not include vaccines against
infectious diseases”. For classification purposes, vaccines are expected to have prophylactic mode
of action, i.e. prevention of an infectious disease in humans. If a product is intended to treat
pathologies caused by the infection (e.g. malignancies), it is classified as a GTMP. Live recombinant
viral vectors (delivering genes encoding specific antigen sequences into human somatic cells) could
fulfil the definition of Gene Therapy Medicinal Products (GTMP) when administered for example in
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
8/19

oncology, but similar products would not be classified GTMPs when intended as prophylactic
against infectious disease. In order to enable the classification of borderline products (treatment of
infections or premalignancies) the therapeutic indication and target population should be clearly
defined.
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
9/19
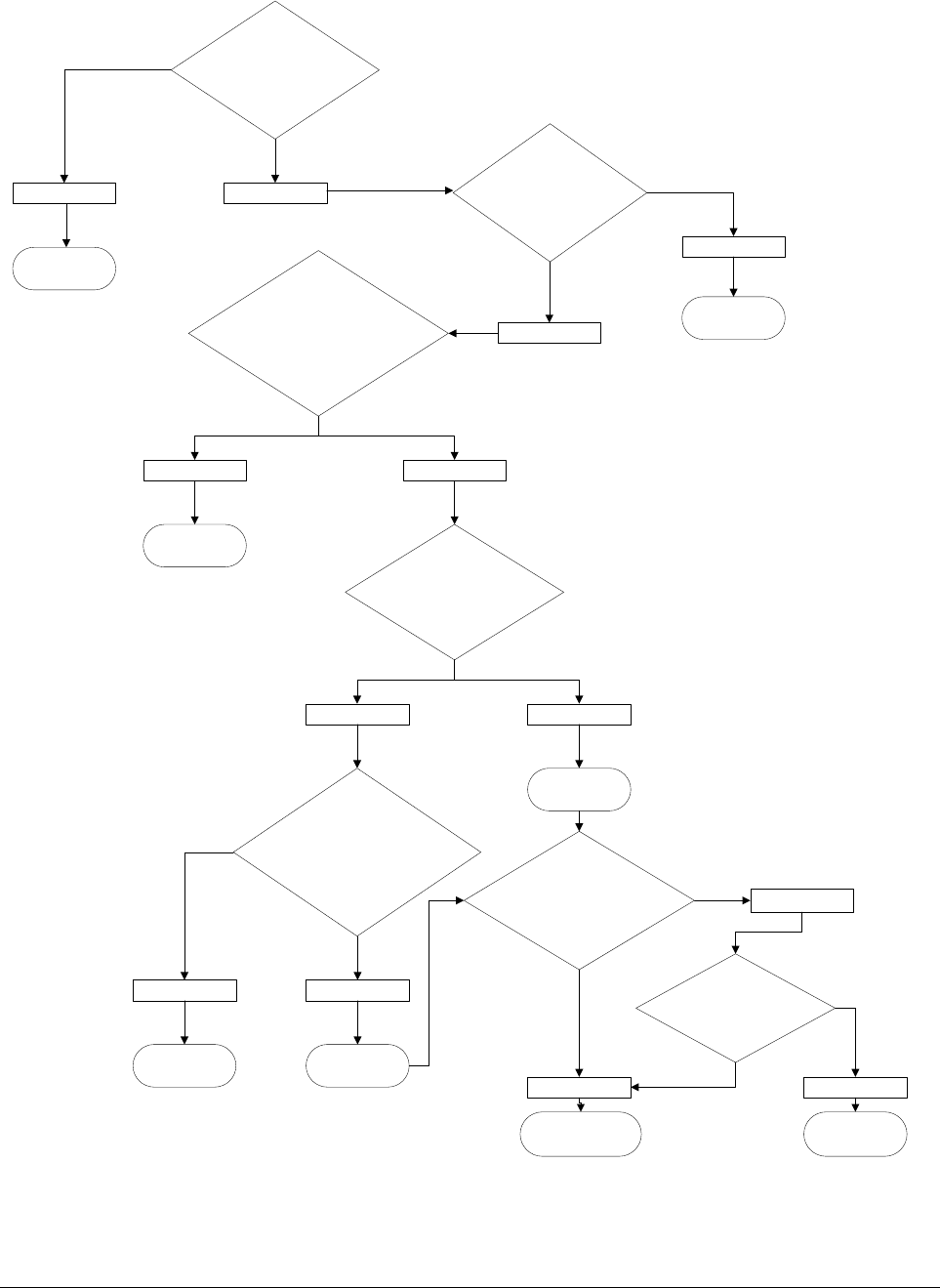
Figure 1. DECISION TREE FOR GTMP
The following questions can help applicants to classify their product:
NO
Biological
active substance contains or
consist a recombinant nucleic
acid
sequence
Not a GTMP
Recombinant
nucleic acid sequence
used in or administered to human being
with a view to regulating,
repairing,
replacing, adding or
deleting a genetic sequence
YES
YESNO
Not a GTMP
Its primary therapeutic,
prophylactic or diagnostic effect
relates directly to the recombinant
nucleic acid sequence
NO
YES
GTMP
Its primary therapeutic
,
prophylactic or diagnostic effect
relates directly to the product of the
expression of the recombinant nucleic
acid sequence
YES
GTMP
NO
Not a GTMP*
Vaccine against infectious
disease
?
YES
Not a GTMP
NO
Does the product contain
(genetically modificed) cells?
bh
Not a combined
ATMP
YES
Combined ATMP
Does the product
contain one or more
medical device as an integral
part of the product
?
Yes
Ex
planatory notes: *) The product can contain genetically modified cells for which specific requirements should be followed (see ‘Guideline on
human cell-based medicinal products’ (EMEA/CHMP/410869/2006).
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
10/19

2.2.4. Criteria for somatic cell therapy medicinal products (sCTMP) and
tissue engineered products (TEP)
a) sCTMP and TEP both contain or consist of engineered cells or tissues (see definition in section 2.1
above). To be considered ‘engineered’, cells or tissue(s) should fulfil at least one of the following
criteria:
• Substantial
manipulation
The cells or tissue(s) have been manipulated during the manufacturing process so that their
biological characteristics, physiological functions or structural properties have been modified to be
relevant for their intended function. Examples of substantial manipulations include cell expansion
(culture), genetic modification of cells, differentiation/activation with growth factors.
Cell culturing leading to expansion is considered substantial manipulation. Induction of proliferation
of cells during cell culture has to be regarded as changes of their biological characteristics and
structural properties, either because of an immediate change in cell functionality or cell phenotype,
or by increasing cell numbers to augment the desired function of the cells. Furthermore, most
adherent cells, for example, are impacted by the repeated attachment and detachment cycles. It
has been demonstrated that even the techniques applied for cell detachment might lead to different
phenotypic changes especially on cell surface proteins (e.g. membrane receptors).
Enzymatic digestion of a tissue to release cells is also considered to be substantial manipulation,
when the aim is to dissociate cell-cell contacts and the released cells are administered into patients
with or without subsequent manipulation. An example would be keratinocytes from skin, for which
enzymatic digestion would destroy the tissue architecture and functional interactions of the cells,
which cannot be regained in the cell suspension: this would be considered as substantial
manipulation.
If the enzymatic digestion leads to isolation of functionally intact tissue units (e.g. pancreatic islets)
or there is scientific evidence that the original structural and functional characteristics are
maintained, the procedure is not considered substantial manipulation.
In case a tissue is treated to remove cells and to be used without any cellular components (e.g.
amniotic membrane, bone) the product is not an ATMP because it does no longer contain cells or
tissue.
If the number of certain cells (e.g. MSCs in fat grafts) is enriched by selection and the processing
does not change the characteristics of the cells, this is not considered a substantial manipulation.
Additionally, based on scientific considerations, the CAT can also consider other manipulations as
“non substantial”. One example is the radiolabelling of leukocytes for diagnostic purposes. This
technique has no significant impact on the functional properties of the cells and should thus not be
considered a substantial manipulation.
• Different essential function (non-homologous use)
.
In case no substantial manipulation of the cells/tissues takes place, the classification is based on
the essential function of the cells/tissues. Such non-substantially manipulated cells or tissues used
for the same essential function are not considered ATMPs. The same essential function for a cell
population means that the cells when removed from their original environment in the human body
are used to maintain the original function(s) in the same anatomical or histological environment.
Examples of this category are bone marrow cells or peripheral blood cells used for haematopoietic
or immune reconstitution. Other clinical uses of bone marrow cells would be considered to be
ATMPs, unless the same essential function(s) and the same anatomical/histological environment can
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
11/19

be demonstrated for the cells/tissues both at the donor and administration site (tissue). The same
principle applies to other non-substantially manipulated cells from various origins, for example
adipose cells transplanted to other than fat tissue are considered to be ATMPs.
Replacement of a tissue as its whole or functional unit of a tissue (such as cornea or pancreatic
islets) is regarded as use for the same essential function. Similarly, transplantation of a non-
manipulated tissue to another location in the same anatomical or histological environment is also
considered to achieve the same essential function. This is the case for skin transplantation from one
part of the body to another part, subcutaneous implantation of pancreatic islets or replacement of
arteria by veins. However, in the case of pancreatic islets, the classification will also depend on the
manipulation and functional integrity of the islets.
b) Differentiation between sCTMP and TEP
The main difference between sCTMP and TEP is determined on the basis of the intended function of the
product as claimed by the Applicant. The sCTMPs are intended for the prevention, diagnosis and/or
treatment of diseases via pharmacological, metabolic actions, whereas TEPs are used in or
administered to human beings with a view to regenerating, repairing or replacing a human tissue. The
decision, whether a product fulfils the requirements of a sCTMP or a TEP, is taken on the basis of the
claimed mode of action in association with its associated claimed intended function.
The therapeutic action of the product i.e. “regeneration– repair – replacement” is an important
component in determining the classification as TEP. These may be interlinked processes that cannot be
defined separately but have to be considered together. The three processes may occur concomitantly
or sequentially (e.g. implantation of chondrocytes to replace missing cartilage followed by repair and
induction of regeneration).
The CAT considers that a product consisting of engineered cells that induces regeneration, repair or
replacement in the native tissue e.g. via secretion of paracrine factors (by the engineered cells/tissue),
also fulfils the definition of a TEP. In many cases, such products would also fulfil the definition of a
sCTMP, and therefore the classification as TEP is based on the demarcation rule in art. 2(4) of the
ATMP Regulation.
Isolated pancreatic beta cells embedded in an alginate matrix may serve as example for the
delineation between somatic cell therapy and tissue engineering: This cell-based product was intended
to be administered to patients with a view to restoring, correcting or modifying physiological function
via a metabolic action of the cells it contains (secretion of insulin). As the claimed MoA of the product
was the transient restoration of beta cell activity (the “replacement of the function”), but not the
regeneration, repair nor the replacement of the human tissue itself, it was concluded that the product
was a somatic cell therapy product. In line with this approach, human liver-derived progenitor cells
were also classified as somatic cell therapy, since the cells serve to primarily replace a function
(treatment of inborn errors of liver metabolism) rather than the tissue itself.
In contrast, a preparation of cells derived from adult skeletal muscle tissue, intended for the treatment
of stress urinary incontinence, was classified as a TEP because the cells were administered primarily
with a view to regenerating, repairing or replacing a human tissue (the replacement of urethral
sphincter muscle cells, or to repair respective injured tissue).
It should be noted that the effect of a tissue engineered product can be transient, e.g. autologous
human keratinocytes intended for the treatment of acute burns may only transiently repair the
underlying structure and later be replaced.
c) Inclusion and exclusions:
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
12/19

- Products containing or consisting of animal cells or tissues to be administered to humans will
always be considered as ATMPs.
- Products containing or consisting exclusively of non-viable cells or tissues and which do no act
principally by pharmacological, immunological or metabolic action, will not be considered
ATMPs.
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
13/19

Figure 2. DECISION TREE FOR sCTMP and TEP
The following questions can help applicants to classify their product:
Product containing cells
or tissues (
human,
animal or both)?
NO
YES
Does the product
contain exclusively
non-
viable cells or
tissue
(
s)
?*
YES
NO
Cells or tissue
(s
)
substantially
manipulated?
YES
NO
Product
intended for
regeneration,
repair
,
replacement of
human tissue
TEP
Product intended for
treatment, prevention
or diagnosis of a
disease through
pharmacological,
immunological or
metabolic action of its
cells / tissues
sCTMP
Go to Gene therapy
flowchart
Not a Gene
therapy medicinal
products
Different
essential
function?
Not a sCTMP nor
a TEP
**
NO
YES
Product intended for
regeneration, repair,
replacement of
human tissue
TEP
Product intended for
treatment, prevention or
diagnosis of a disease
through pharmacological,
immunological or
metabolic action of its
cells
/ tissues
sCTMP
non-viable
cells
or tissues
act principally by
pharmacological,
immunological or
metabolic action?
YES
NO
Not a sCTMP nor
a TEP
Does the product contain one or
more Medical devices/
active
implantable medical device as an
integral part of the product?
NO
Not a
combined
ATMP
YES
Combined ATMP
Not a sCTMP nor
a TEP
Cells genetically
modified
?
YES
NO
Gene therapy
medicinal products
Explanatory notes:
*) see section 2.2.1 on what are considered viable cells. It should be noted that a product containing
exclusively non-viable cells/tissue and a medical device / active implantable medical device as an
integral part, will be considered a combined ATMP when these non-viable cells/tissues exert the
primary action of the combined product. This primary action should be based on the pharmacological,
immunological or metabolic action of the non-viable cells/tissues.
**) See section 2.2.4. Criteria for somatic cell therapy medicinal products (sCTMP) and tissue
engineered products (TEP)
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
14/19

2.2.5. Criteria for combined ATMPs
A product is classified as a combined ATMP when it fulfils the definitions provided in Article 2(1)(d) of
the ATMP Regulation (EC) No 1394/2007 (See Section 2.1 above).
Combined ATMPs incorporate an active substance, i.e. a cellular or tissue part consisting of viable or
non-viable cells or tissues and of one or more medical devices or one or more active implantable
medical devices as an integral part of the product. The medical device or active implantable medical
device(s) should be used in the combination in the same way as its intended use without additional
components. If cells or tissues are not viable these must exert the primary action of the combined
product.
Examples of combined ATMPs:
• The expanded autologous chondrocytes seeded onto a collagen membrane and administered, fixed
on this membrane, into the joint cartilage lesion. The primary action of the combined product is
given by the viable cells that repair the damaged tissue, while the medical device part is a tool that
is needed to retain the cells physically to the cartilage defect.
• Autologous osteoprogenitor cells, isolated from bone marrow, are grown within and around a
bioresorbable scaffold that acts as physical support. The finished combined product is an integrated
product consisting of a cellular component and a matrix. The repairing/replacing effect on the bone
defect is accomplished by the living cells that continue to grow within the lesion while the
biodegradable matrix is gradually eliminated. However, like in the first example, the matrix still has
its intended function at the time of implantation.
• Genetically engineered cells - where a recombinant human gene in a mammalian expression vector
is introduced into human cells through transfection and resulting cells are further cultured in vitro -
incorporate as an integral part of the product two components, a semipermeable hollow fibre
membrane (HFM) capsule and a scaffold of strands of polyethylene terephthalate (PET) yarn. Both
components fulfil the definition of medical devices and/or active implantable medical devices as
they are required for maintenance of the cells (growth support, delivery of nutrients) and the
semipermeable capsule is needed for release of the therapeutic molecule. As the combined product
fulfils both definitions of a tissue engineered product and a gene therapy medicinal product, it was
classified as a combined gene therapy medicinal product.
It should be noted that normally the medical device should retain its intended purpose / mode of action
in the combination to be considered as being “integral part” of the final product and thus qualify this
product as a combined product. The CAT has therefore classified some products as non-combined
ATMP, where the function of the matrix was no longer considered to be linked to its structural
properties. This is discussed further in section 2.3.4 on Combined ATMPs versus non-combined ATMPs.
2.3. Evolving and borderlines areas
The ATMP classification procedure will also have to clarify borderline cases between ATMPs versus non-
ATMPs as well as between the different product categories within the ATMP sphere. Below are given
examples that illustrate the type of issues that are taken into consideration when assessing borderline
cases.
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
15/19

2.3.1. Advanced therapies versus transplants/transfusion
Products consisting of cells or tissues may scientifically be at the border between Tissues and Cells
directive (Directive 2004/23/EC) and the ATMP regulation. Cells/tissues harvested and separated by a
simple selection method (that does not result in a substantial manipulation of the cells/tissue) and re-
administered to fulfil their same essential function will generally be regarded as non-ATMPs. However,
depending on whether or not the selection process/method will alter the original characteristics of the
cells/tissues may result in classification as ATMPs. Similarly, cells derived from human blood (e.g.
lymphocytes) that are substantially manipulated or use for a different essential function are classified
as ATMPs.
One example is the recommendation of the CAT that a preparation of human pancreatic Langerhans’
islets should not be classified as an ATMP. The CAT considered that, for this preparation, the described
process steps do not constitute substantial manipulations for the intended use so that there is no
change in the biological characteristics of the islets. In addition, the product was intended to be used
for the same essential function in the recipients, be it in the allogeneic or autologous conditions
described. This conclusion is, however, not directly applicable to any other pancreatic beta cell
products which may be submitted for classification, as they may be derived from very different and
more complex process and substantial manipulations, as discussed also in section 2.2.4 (cell-based
product consisting of isolated beta-cells embedded in an alginate matrix).
In contrast, some products previously considered as non-ATMP because of an essentially minimal
manipulation or maintenance of the initial biological properties have been classified as ATMP due to
their intended use based on (a) different essential function(s) of the cells/tissues. For example, the use
of autologous bone marrow-derived progenitor cells intended for treatment of patients with myocardial
infarction or other vascular diseases would be considered as different essential function and therefore
such products are classified as ATMPs (in this case tissue engineering products) (see section 2.2.4).
It is possible that cell-based products administered in the same anatomical location fall under the
definition of ATMP on grounds that it is used for a different essential function. This can be encountered
when the mode of action of the cells is not identical to the one attributed to the cells by the scientific
knowledge, for example, the injection of concentrated bone marrow at the site of bone injury with the
aim of healing a bone lesion.
2.3.2. Classification of genetically modified bacteria as Gene therapy
medicinal product
The CAT has discussed several examples of genetically modified bacteria which express a human gene
sequence in the patient after administration. These products raised difficult questions about the
interpretation of the first indent in the definition of gene therapy medicinal product (i.e. that “it
contains an active substance which contains or consists of a recombinant nucleic acid used in or
administered to human beings with a view to regulating, repairing, replacing, adding or deleting a
genetic sequence” ). The following considerations are relevant in this regard: (i) it could be considered
that the genetic sequence is not “added” to human cells, but remains in the bacteria, and equally also
the protein it expresses; and (ii) it could be considered that the medicinal product is adding a genetic
sequence into humans to elicit a pharmacological effect. However, given that the first indent of the
definition does not include the requirement that the repair, replacement, addition or deletion of the
genetic sequence is done “at the level of the human cell”, the CAT classified this medicinal product as a
gene therapy medicinal product. The consideration that prevailed was therefore that a genetic
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
16/19

sequence is administered to humans and that the effect is due to the product expressed from this
added genetic sequence.
2.3.3. Gene therapy medicinal product versus cell therapy medicinal
product
Another borderline scenario relates to products that are modified by adding a mRNA sequence, for
example dendritic cells (DC) electroporated with mRNA in vitro and administrated to the patient to
elicit a specific immune response. One could argue that the claimed mechanism of action is directly
related to the expression of the mRNA encoded antigens to stimulate e.g. tumour specific immune
responses. However, due to its relatively short half-life there may be little or no residual mRNA at the
time of re-administration of the dendritic cells to the patient. Thus, it can be claimed that a
recombinant nucleic acid is not administered to human beings with a view to adding a genetic
sequence, but rather the mRNA electroporated DCs could be seen as an intermediate in the
manufacturing process where the phenotype is finally altered without alteration of the genotype of the
cells. Therefore, the product was considered not to comply with the definition of a gene therapy
medicinal product. Instead the CAT considered that the product was a somatic cell therapy product as
it consists of cells which were administered to human beings with a view to treating a disease through
the immunological action of the modified cell populations.
2.3.4. Combined ATMPs versus non-combined ATMPs
The border between combined or non-combined ATMPs is often discussed in classification procedures.
This section should be read in conjunction with the section 2.2.5, which describes the criteria for
combined ATMPs. In this regard it is relevant to consider if (i) the medical device is an integral part of
the final product (combined) or (ii) if the combined component (although CE marked) is not or no
longer used as a medical device but should be considered as an “excipient” in the final formulation of
the drug (and therefore not combined).
Below are two examples where the matrix does not retains its original intended structure and/or does
no longer function as a medical device and one example where the collagen matrix is acting as a
medical device.
• An example of non-combined ATMP can be given with the human endothelial cells cultured in a
gelatin matrix and used to treat vascular injury. The product was claimed to reduce the intimal
thickening of vessels injured by the frequent procedures of artero-venous grafts and fistula
placements in patients that undergo haemodialysis. The underlying mechanism of action is based
on the concept that the allogeneic endothelial cells release biological factors that inhibit the
intimal hyperplasia, reduce the graft thrombosis, and repair the vascular injury. The gel matrix is
a CE marked medical device indicated in surgical procedures as an adjunct to haemostasis. The
gel, which is seeded with the cells, contributes to the formulation of the final product. The gel
matrix has the function to keep the cells around the vascular injury site to release the therapeutic
factors, but it is also contributing in some way to provide the correct signals to the cells. The CAT
considered that the porcine gelatine matrix, as a component of this medicinal product, is
remodelled by the cells contributing to product efficacy. Thus, the manufacturing process uses
the matrix in a different way than its intended use when considered as a medical device. In this
formulation (e.g. the porcine gelatine matrix and the human aortic endothelial cells), the matrix
was not considered to be a medical device any more. The CAT therefore classified the product as
a sCTMP, not combined ATMP.
• A similar situation applies to another example, which is the mixture of pancreatic beta cells and
their accompanying endocrine cell populations embedded in an alginate matrix intended for the
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
17/19

treatment of diabetes. The CAT was of the opinion that the inert alginate matrix is reworked by
the cells during culture and becomes an integral part of the product that supports to
contain/preserve the biological characteristics and functional activities of the cells. The function of
the alginated matrix was no longer considered to be linked to its structural properties. The CAT
therefore classified the product as a sCTMP, not combined ATMP.
• In contrast, human fibroblasts cultured onto a biodegradable collagen matrix were classified as a
TEP, combined ATMP. Here, the matrix is an integral part of the product and it fulfils its function
as CE marked medical device when administered to patients.
2.4. Clarifications on procedural aspects information to be submitted by
the applicant
In order to facilitate the access to the ATMP classification, the CAT has published the procedural
advice for the ATMP classification
5
, which describes the procedure and gives guidance for the steps
to be followed by the applicant for the submission of an ATMP classification.
Upon receipt of a valid request
6
, the CAT delivers a scientific recommendation on an ATMP
classification after consultation with the European Commission within 60 days.
Sufficient scientific information relevant to the decision is essential to be submitted in order for the
CAT to classify a product, e.g. on following areas:
• Active substance: description of active substance (including starting materials, when relevant), any
additional substances (e.g. when applicable: structural component such as scaffolds, matrices,
biomaterials, biomolecules and/or other components), medical device or active implantable medical
device (including information on the classification status of the Medical Device from a Medical
Device Competent Authority when applicable).
• Finished Product: qualitative and quantitative (where available) composition, mode of
administration, pharmaceutical form and description of the finished product ready for clinical use.
• Mechanism of Action/ Proposed use: claimed mechanism of action, properties (including
pharmacological, immunological or metabolic, if applicable), proposed use / indication (including
therapeutic, prophylactic, diagnostic). See also section 2.2.1. above. Applicants should provide an
in-depth discussion on how the product works and what data are available to support the
mechanism of action. This is essential, since the outcome of the classification will depend on the
claim the Applicant provides and how strong the evidence is to support it. For example, the CAT
was for one product not able to classify it as tissue engineered product or somatic cell therapy
medicinal product, since the claim for the mechanism of action was not sufficiently defined, and not
enough data (be it data with the product or what is published for that given product class) was
presented to support the Applicant’s claims.
• Summary of the status of the development of the product: key elements of manufacturing, quality
aspects (including description and level of manipulations on cells and tissues, when applicable).
Outline of Non-Clinical development and Clinical development relevant for the ATMP classification.
5
Procedural advice on the provision of scientific recommendation on classification of advanced therapy medicinal
products in accordance with Article 17 of Regulation (EC) No 1394/2007
6
For the submission of an ATMP classification, applicants should complete a Pre-submission request form (selecting
in the drop-down menu ATMP-ATMP classification) and the ATMP Classification Request form and briefing
information and return both to: AdvancedTherapies@ema.europa.eu
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
18/19

Depending on the stage of development at which the classification advice is sought, some of the
parameters or information requested above may not be finalised. In this case, the target profile
and intended product description may suffice.
In addition to the qualitative and quantitative description of the product to be classified, applicants are
encouraged to present their views on the classification of products under development. They should
discuss any aspects supporting or not the applicability of the pharmaceutical framework for the
development and evaluation of the product. Overlapping aspects relevant to medical devices,
cosmetics, human tissues and cells, blood products, borderline medical use or other issues should also
be highlighted if appropriate.
Details of the regulatory status of the product (including medical device/active implantable device,
when applicable), marketing history in EU and non EU countries and information on the current
medical use worldwide are requested to complement the overall understanding on the regulatory
status of the candidate ATMP.
Applicants can include in the request any additional information or bibliographic references to further
substantiate their positions on the classification of their product on the light of legal definitions in force.
i
Article 17(1) of the ATMP Regulation: Any applicant developing a product based on genes, cells
or tissues may request a scientific recommendation of the Agency with a view to determining
whether the referred product falls, on scientific grounds, within the definition of an advanced
therapy medicinal product. The Agency shall deliver this recommendation after consultation with
the Commission and within 60 days after receipt of the request.
(2). The Agency shall publish summaries of the recommendations delivered in accordance with
paragraph 1, after deletion of all information of commercial confidential nature.
ii Directive 2001/83/EC Annex I Part IV: Web link to Directive 2009/120/EC:
http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2009:242:0003:0012:EN:PDF
iii
Recital 24 of ATMP Regulation: The Agency should be empowered to give scientific recommendations
on whether a given product based on genes, cells or tissues meets the scientific criteria which define
advanced therapy medicinal products, in order to address, as early as possible, questions of borderline
with other areas such as cosmetics or medical devices, which may arise as science develops. The
Committee for Advanced Therapies, with its unique expertise, should have a prominent role in the
provision of such advice.
iv A medicinal product as defined in Article 1(2) of Directive 2001/83/EC, as amended, is:
(a) Any substance or combination of substances presented as having properties for treating or
preventing disease in human beings;
or
(b) Any substance or combination of substances which may be used in or administered to human
beings either with a view to restoring, correcting or modifying physiological functions by exerting a
pharmacological, immunological or metabolic action, or to making a medical diagnosis"
A biological medicinal product as defined in section 3.2.1.1 (b) of the Annex I to Directive 2001/83/EC:
A biological medicinal product is a product, the active substance of which is a biological substance.
A biological substance is a substance that is produced by or extracted from a biological source and that
needs for its characterisation and the determination of its quality a combination of physicochemical-
biological testing, together with the production process and its control. The following shall be
considered as biological medicinal products: immunological medicinal products and medicinal products
derived from human blood and human plasma as defined, respectively in paragraphs (4) and (10) of
Article 1; medicinal products falling within the scope of Part A of the Annex to Regulation (EEC) No
2309/93; advanced therapy medicinal products as defined in Part IV of this Annex.
Reflection paper on classification of Advanced Therapy Medicinal Products
EMA/CAT/600280/2010 rev.1
19/19
