Page 1 AP Biology: 2013 Exam Review
AP BIOLOGY EXAM REVIEW GUIDE
“The price of success is hard work, dedication to the job at hand, and the determination that
whether we win or lose, we have applied the best of ourselves to the task at hand.”

Page 2 AP Biology: 2013 Exam Review
CONCEPT 1 - BIOCHEMISTRY
1. CHNOPS- most common elements in all living matter
2. Bonds- ionic (transfer electrons), covalent (sharing- polar/unequal sharing and non-polar/equal sharing),
hydrogen (weak bonds between hydrogen and negatively charged items), hydrophobic interactions (how
non-polar compounds congregate together- lipids)
3. pH
a. acid-base/ 0-14, # of H ions determines scale; logarithmic- pH 3 = 10
-3
= 1/1000
b. blood- 7.4, stomach- 2, small intestine- 8; enzymes are specific to pH
4. Water properties- polarity, cohesion(attraction to other water molecules), adhesion (attraction to other
charged compounds) low density when frozen, versatile solvent, high heat of fusion/vaporization;
surface tension
5. Organic molecules - monomers are simplest form of all; monomers join together via dehydration synthesis
(loss of water) to make polymers; polymers are broken down via hydrolysis (input of water)
a. Carbohydrates- CHO 1:2:1 ratio, monomer= monosaccharides, 2=disaccharides, 3 or more=
polysaccharides
Used for energy (cell respiration)
Examples
(1) glucose- immediate energy to make ATP
(2) starch- stored energy in plants
(3) glycogen- stored energy in animals (stored in liver)
(4) cellulose- plant cell wall
b. Lipids – C, H, O (not a 1:2:1 ratio) *P only in phospholipids
(1) fats, waxes, oils and sterols
(2) Saturated fats have single bonds between carbons, unsaturated fats have at least one
double bond between carbons (kinky); plants make polyunsaturated; animals make
monounsaturated
(3) Phospholipids make up cell membranes (double layer) and are amphipathic- hydrophilic and
hydrophobic
(4) Uses- in all membranes; stored energy, protection, insulation, myelin sheath of nerves
c. Proteins- C, H, O, N (may have other elements in R group)
(1) Monomer- amino acids (20 total types), 2=dipeptide, 3 or more= polypeptide
(2) Parts of amino acid= carboxyl group (COOH) on one end, amino group on the other end
(NH2), central carbon and variable R group (can be hydrophobic or hydrophilic) which
determines chemical properties.
(3) Protein Folding- shape determines function; primary= a.a. chain; secondary= beta pleated
sheet or alpha helix( hydrogen bonds); tertiary=globular; folds in on itself (disulfide bridges,
hydrogen bonds, hydrophobic interactions; ionic bonding); quartenary= more than one
polypeptide.
(4) Uses- protein carriers in cell membrane, antibodies, hemoglobin, enzymes, most hormones

Page 3 AP Biology: 2013 Exam Review
d. Nucleic acids – C, H, O, N
(1) Monomer= nucleotide, 2 = dinucleotide, 2 or more polynucleotide
(2) Nucleotide made up of sugar, phosphate and base
(3) Used to store genetic information
(4) DNA is double stranded, has deoxyribose, A, G, C, T
(5) RNA is single stranded, has ribose, A, G, C, U
(6) mRNA- copies genetic message; rRNA- attaches mRNA and makes up ribosomes (most
common);tRNA- carries amino acids; DNA- carries genetic code
6. Enzymes
a. Biological catalysts (made of protein) that speed up rate of chemical reactions by lowering
activation energy required for reaction to occur
b. Enzyme has active site (exposed R groups) where reaction occurs
c. Enzymes can break down substance (catabolic reaction) or build up substances (anabolic)
d. Enzyme/substrate complex is formed
e. Substrate is what enzyme acts on
f. Rate is determined by collisions between substrate and enzyme
g. Ends in –ase, named after substrate often
h. Enzyme is specific to substrate; the substrate must be complementary to the surface properties
(shape and charge) of the active site (which is made up of R groups with specific chemistry, i.e.
hydrophobic).
i. Enzyme rate is affected by:
pH (optimal for each enzyme),
temperature (optimal for each enzyme but in general increased temp means increased
collisions so rate goes up initially; too much heat can denature enzyme), enzyme
concentration (more enzyme faster rate or vice versa)
substrate concentration (more substrate faster rate; v
max
is fastest enzyme can work when
saturated)
j. Inhibition-competitive inhibition (something competes for active site; can be overcome with more
substrate)
k. Non-competitive inhibition- attaches at allosteric site and changes shape of enzyme so it is not
functional; can not be overcome with more substrate
l. Coenzymes (organic; NAD and vitamin B etc.) and cofactors (inorganic; zinc, magnesium etc.)
interact with enzymes to put them into the right structure to do work.
Vocabulary
active site
allosteric site
amino acid
amphipathic
anabolic
carbohydrate
carbon
catabolic
catalyst
coenzyme
denaturation
disaccharide
hydrogen bond
hydrophilic
hydrophobic
ion
lipid
macromolecule
monomer
monosaccharide
non-polar molecule
nucleic acid
nucleotide
organic molecule
peptide bond
polar molecule
polymer
protein
substrate
water

Page 4 AP Biology: 2013 Exam Review
Thinking Practice
1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would
result? Circle the parts of each amino acid that will interact and draw the resulting molecule.
2. Construct a bar graph that displays the relative amounts of hydrogen, carbon, oxygen, and nitrogen in
each of the four types of macromolecules (carbohydrates, lipids, proteins, nucleic acids).
3. Describe the relationship between substrate concentration and reaction rate shown in the graph below and
propose an explanation for it.

Page 5 AP Biology: 2013 Exam Review
4. DNA polymerase from T. aquaticus (Taq) is used in PCR (polymerase chain reaction). PCR is a technique
where millions of copies of DNA can be made from one original copy. In this method, the target DNA
molecule is subjected to temperatures over 95 °C to make the double-stranded DNA separate. The
temperature is then lowered slightly to allow primers to anneal before the Taq polymerase catalyzes the
reactions to incorporate new nucleotides into the complementary strands. The cycle is then repeated over
and over until there are millions of copies of the target DNA.
a. Predict why this bacterial polymerase is used instead of a human polymerase.
b. What would happen if you used a human polymerase in a series of PCR reactions?
5.

Page 6 AP Biology: 2013 Exam Review
Biochemistry – Short Free Response (4 points)
Water is importan t for all living organisms. The functions of water are directly related to its physical
properties. Describe how the properties of water contribute to TWO of the following:
Transpiration
thermoregulation in endotherms
plasma membrane structure

Page 7 AP Biology: 2013 Exam Review
CONCEPT 2 - CELLS
1. Prokaryotic (Bacteria) Eukaryotic (all other living things)
no membrane-bound organelles m.b.o, ex. Chloroplasts and nucleus
no nucleus(single; circular DNA) multiple linear DNA
free ribosomes and cell wall histones on DNA
2. Cell organelles
a. Nucleus- holds DNA and nucleolus(where ribosomal subunits are made)
b. Mitochondria- double membrane; outer is smooth and inside is folded with enzymes to make ATP
(site of cellular respiration (glucose breakdown)
c. Ribosome- site of translation- protein synthesis; made of rRNA and protein
d. E.R.- connected to nucleus; allows for reactions, membranous; smooth= lipids; rough=proteins
e. Golgi complex- packaging in membrane and signals for export
f. Cytoskeleton: Microfilaments- contractile protein, gives shape, movement within cell;
Microtubules- centrioles, cilia, flagella, spindle fibers
g. vacuoles/vesicles- water and solutes; large and central in plants
h. ANIMAL
Lysosomes- contain enzymes; used for intracellular digestion and apoptosis
Centrioles- used in cell division
i. PLANT
Chloroplast- double membrane; site of photosynthesis (glucose synthesis)
Cell wall- middle lamella- pectin; primary cell wall- cellulose; secondary cell wall- lignin
j. Endosymbiont theory- all eukaryotic cells came from bacterial cells that lived together; proof= all
chloroplasts and mitochondria have own DNA and are autonomous
3. Cell membrane (separates the internal environment of cell from external environment).
a. Phospholipid bilayer (selectively permeable; amphipathic)
b. Fluid mosaic model (in motion; proteins, cholesterol, glycoproteins and glycolipids among
phospholipids). Membrane is hydrophilic on inside and outside, hydrophobic within membrane
c. Simple diffusion- from high to low concentration- small and uncharged move freely through
phospholipids ex. CO
2,
O
2
(passive; no energy; no protein carrier)
d. Facilitated diffusion- large or charged from high to low, passive; with protein carrier: ex. glucose, K+
e. Active transport- from low to high concentration; uses ATP; uses a protein
f. Endocytosis- phagocytosis (solid) and pinocytosis (liquid); membrane surrounds and forms vesicles;
receptor mediated endocytosis has receptors on surface
g. Exocytosis- release of material using vesicles fusing with membrane
h. Osmosis- diffusion of water using a selectively permeable membrane; passive; no proteins
i. Water potential= pressure potential plus pressure potential; water moves from high water
potential to low water potential; solutes always lower water potential; pressure can increase or
decrease depending on if it is negative or positive.
j. Plant cells have pressure related to cell wall and vacuole; turgor pressure
k. Hypertonic (high solute), hypotonic (low solute), and isotonic solutions(equal concentration)
l. High surface area : volume ratio increases rate at which food can be taken in a waste expelled
Page 8 AP Biology: 2013 Exam Review
4. Nervous System
a. function: sensory input, motor function, regulation
b. structure: neuron, axon, dendrites, synapse
c. Polarized neuron: Na
+
outside, K
+
and Cl
-
inside
d. Depolarization moves Na into neuron, generating an action potential
e. Repolarization exchanges Na
+
and K
+
through the sodium-potassium pump
f. At synapse, calcium channels open to allow calcium to rush in, stimulating release of
neurotransmitters
g. Neurotransmitters released into synapse to generate action potential for motor neuron or muscle
cell
Vocabulary
active transport
amphipathic
apoptosis
aquaporin
axon
carrier protein
cell wall
centrioles
channel protein
chloroplast
concentration gradient
cytoplasm
cytoskeleton
dendrites
depolarization
diffusion
endocytosis
endoplasmic reticulum
Golgi apparatus
hypertonic
hypotonic
isotonic
ligand
lysosome
membrane
mitochondrion
neuron
neurotransmitter
nuclear envelope
phospholipid
plasma membrane
plasmolysis
polarization
prokaryotic cell
repolarization
ribosome
rough ER
selectively permeable
smooth ER
synapse
exocytosis
eukaryotic cell
facilitated diffusion
flagella
fluid mosaic model
nucleus
organelles
osmosis
passive transport
phagocytosis
surface area:volume ratio
transmembrane protein
vacuole

Page 9 AP Biology: 2013 Exam Review
Thinking Practice
1. For each molecule shown to the
right, answer the following,
providing justifications for each:
a. Is it polar or nonpolar?
b. Is it hydrophobic or
hydrophilic?
c. In order to be transferred
into a cell, would the
molecule require a protein
channel?
2. Biological systems rely heavily on the properties of water movement. Excretion, digestion, and blood
pressure are just a few examples of situations where water balance is important. Suppose you have a
semi-permeable membrane that ONLY water can pass. On one side of the membrane you have 0.1 M
CaCl
2
. On the other side of the membrane, you have 0.1 M Glucose. CaCl
2
ionizes in water to produce 3
ions. Glucose does not ionize in water.
0.1 M CaCl
2
0.1 M Glucose
a. Calculate the water potential for each side of the membrane.
b. Describe which way water will move and explain your answer.

Page 10 AP Biology: 2013 Exam Review
4. The following diagram shows an action potential of a neuron. For
each question, you can answer with one letter or multiple letters.
a. At which letters would you find Na+ voltage gated channel
OPEN?
b. At which letter(s) would you find the Na+/K+
pump WORKING?
c. At which letter(s) would you find K+ voltage gated channels
OPEN?
d. At point F, would there be a more positive charge on the INSIDE or OUTSIDE of the neuron?
e. At point B, would you find more Na+ on the INSIDE or OUTSIDE of the neuron?
f. Tetrodotoxin is a neurotoxin that blocks Na+ voltage gated channels. How would the function of
the neuron be altered by the presence of this toxin?
5. Tay-Sachs disease is a human genetic abnormality that results in cells accumulating and becoming clogged
with very large and complex lipids. Which cellular organelle must be involved in this condition?
3.

Page 11 AP Biology: 2013 Exam Review
Cells – Long Free Response (10 points)

Page 12 AP Biology: 2013 Exam Review

Page 13 AP Biology: 2013 Exam Review

Page 14 AP Biology: 2013 Exam Review
CONCEPT 3 – ENERGY AND METABOLISM
1. Energy
a. Organisms use free energy for organization, growth and reproduction. Loss of order or free energy
flow results in death.
b. More free energy (ex. Food) than needed will be stored for growth (roots, glycogen, fat, etc.).
c. Matter and energy are not created but change form (1
st
law of thermo; ex. Sun energy to bond
energy in glucose) and entropy is increasing in disorganization of energy (i.e. heat released by cell
respiration). More organized or built up compounds have more free energy and less entropy (i.e.
glucose) and less organized have less free energy and more entropy (i.e. carbon dioxide).
d. Reactions can be coupled to maintain a system, ex. Photosynthesis and cell respiration
2. Cellular respiration C
6
H
12
O
6
+ 6O26CO
2
+ 6H
2
O
a. Makes ATP for cell use; uses glucose and oxygen makes waste products of carbon dioxide and
water; occurs in mitochondria; NADH is electron carrier used
b. Glycolysis
(1) occurs in cytoplasm; anaerobic
(2) rearranges the bonds in glucose molecules, releasing free energy to form ATP from ADP
through substrate-level phosphorylation resulting in the production of pyruvate.
c. Kreb’s cycle
(1) occurs in mitochondrial matrix
(2) also called the citric acid cycle
(3) occurs twice per molecule of glucose
(4) Pyruvate is oxidized further and carbon dioxide is released ; ATP is synthesized from ADP
and inorganic phosphate via substrate level phosphorylation and electrons are captured by
coenzymes (NAD+ and FAD).
(5) NADH and FADH2 carry electrons to the electron transport chain.
d. Electron Transport Chain and Chemiosmosis
(1) The electron transport chain captures electrons, pumping H
+
ions into the inter-membrane
space of the mitochondria.
(2) Electrons are accepted by O
2
molecule forming H
2
O
(3) Concentration of H
+
builds up within inter-membrane space lowering the pH and ions rush
through ATP synthase into the mitochondria matrix. Rush of ions “spins” ATP synthase
protein, causing ADP and P
i
to join forming ATP by oxidative phosphorylation
3. Photosynthesis 6CO
2
+ 6H
2
O C
6
H
12
O
6
+ 6O
2
a. Photosynthetic organisms capture free energy present in sunlight and use water and carbon
dioxide to make carbon products and free oxygen.
b. Light-dependent reactions- photophosphorylation
(1) Photosystems I and II (chlorophyll and proteins) are embedded in the internal membranes
of chloroplasts (thylakoids of the grana). They pass electrons through an electron transport
chain (ETC). When electrons are passed they allow hydrogen ions (protons) across the
thykaloid membrane. The formation of the proton gradient powers the process of ATP
synthesis to add a phosphate ADP to ATP (chemiosmosis).
Page 15 AP Biology: 2013 Exam Review
(2) Electrons are passed to NADP+ to make NADPH (electron carrier)
(3) H
2
O is used and O
2
released as by-product
(4) Red and blue light works best (green is reflected typically)
(5) Energy converted from sun into chemical energy of ATP and NADPH to be used in building
of sugar (Calvin Cycle)
c. Light-independent reactions- Calvin Cycle
(1) carbon fixation occurs (carbons of CO
2
used to make sugar)
(2) occurs in stroma of chloroplasts
(3) ATP and NADPH generated by light-dependent reactions are used to assemble glucose
4. Anaerobic Fermentation
a. No oxygen; cell only goes through glycolysis followed by fermentation
b. Fermentation recycles NAD needed to restart glycolysis
c. alcohol fermentation ex. yeast cells- glucose ethyl alcohol + CO
2
+ NAD
+
d. lactic acid fermentation ex. muscle cells- glucose lactic acid + NAD
+
e. Fermentation does not make ATP but glycolysis does- 2ATP; very inefficient; sufficient for
microorganisms
Vocabulary
absorption spectrum
acetyl coA
anabolism
anaerobic metabolism
ATP
ATP synthase
autotroph
Calvin cycle
cellular respiration
chemiosmosis
chlorophyll
chloroplast
citric acid/Krebs cycle
electron transport chain
FAD/FADH
2
feedback inhibition
fermentation
glycolysis
light dependent reactions
light independent reactions
metabolic pathway
mitochondrion
NAD/NADH
NADP/NADH
oxidative phosphorylation
photolysis
photosynthesis
photosystem I
photosystem II
pyruvate
stroma
substrate-level phosphorylation
thylakoid membrane

Page 16 AP Biology: 2013 Exam Review
Thinking Questions
1. The figure below outlines the process of cellular respiration. Glucose and oxygen are both reactants in this
process.
a. Describe the journey of a single carbon atom from glucose in cellular respiration
b. Describe the journey of a single hydrogen atom from glucose in cellular respiration
c. Describe the function of the oxygen molecules in cellular respiration

Page 17 AP Biology: 2013 Exam Review
2. The figure below outlines the process of photosynthesis. Carbon dioxide and water are both reactants in
this process.
a. Describe the journey of a single hydrogen atom from water in photosynthesis.
b. Describe the journey of a single oxygen atom from water in photosynthesis.
c. Describe the journey of a carbon dioxide molecule in photosynthesis.

Page 18 AP Biology: 2013 Exam Review
3. It is estimated that more than 2 × 1026 molecules of ATP are hydrolyzed in the human body daily. If each
molecule was used only once you would need approximately 160 kg (350 lbs) of ATP daily. The repeated
use of ATP molecules through the ATP cycle saves the body a huge amount of resources and energy.
ATP is synthesized in two ways:
Substrate-level phosphorylation—Energy released during a reaction, such as the breakdown of sugar
molecules, is used directly to synthesize ATP. A small amount of energy is generated through this
process.
Electron transfer (oxidative phosphorylation)—Energy from the movement of electrons from one
molecule to another, via electron carriers, is used to synthesize ATP. Most cellular ATP is synthesized
by electron transfer in the mitochondria.
Dinitrophenol (DNP) is an “uncoupler,” which means it interferes with the flow of electrons during electron
transfer. Fifty years ago, DNP was given as a drug to help patients lose weight.
a. Why would taking DNP make someone lose weight?
b. Why would taking DNP be dangerous?
4. An experiment to measure the rate of respiration in crickets and mice at 10
o
C and 25
o
C was performed
using a respirometer, an apparatus that measures changes in gas volume. Respiration was measured in mL
of O
2
consumed per gram of organism over several five-minute trials and the following data were
obtained.
a. Which organism at which temperature had the fastest metabolic rate (produced the most ATP)
during its trials? Explain how you know.
b. According to the data, the mice at 10
o
C demonstrated greater oxygen consumption per gram of
tissue than did the mice at 25
o
C. Propose an explanation for why this is.

Page 19 AP Biology: 2013 Exam Review
5. Under laboratory conditions, muscle cells were broken up and separated into fractions of mitochondria
and cytoplasm in an attempt to learn more about cellular respiration. Each fraction was incubated with
glucose or pyruvate. Tests were carried out during incubation for the presence of either carbon dioxide or
lactic acid. The results are shown below:
a. What does the presence of lactic acid in a sample indicate about what process is occurring in each cell
fraction?
b. Explain why lactic acid was produced by the cytoplasm fraction incubated with glucose, but not the
mitochondrial fraction.
c. Why was no carbon dioxide produced by either fraction incubated with glucose?
d. Why did the cytoplasm fraction produce lactic acid in the presence of both glucose and pyruvate?
e. Why did the mitochondria produce carbon dioxide in the presence of pyruvate but not in the presence
of glucose?
6. The figures to the right display the absorption range for several
different pigments found in plants (top) and the rate of
photosynthesis at varying conditions of wavelength in one plant
species (bottom):
a. What color and wavelength of light is reflected by the plant
species tested? How do you know?
b. What wavelength(s) increase the rate of photosynthesis in
the plant species tested? What pigment does this
correspond to? How do you know?

Page 20 AP Biology: 2013 Exam Review
Energy and Metabolism Short Free Response (4 points)
In a second experiment, Variety A seedlings at 17
o
C were treated with a chemical that prevents NADH from
being oxidized to NAD+. Predict the most likely effect of the chemical on metabolism and oxygen consumption
of the treated seedlings. Explain your prediction.

Page 21 AP Biology: 2013 Exam Review
CONCEPT 4 – THE CELL CYCLE AND HEREDITY
1. Cell cycle
a. Reason for division- as cells increase in volume, the surface area decreases and demand for
material resources increases which limits cell size
b. Smaller cells have a more favorable surface area-to-volume ratio for exchange of materials with the
environment (diffusion, etc.). High SA:V ratio is favorable. Ex. 6:1 is better than 6:5
c. Cell cycle switches between interphase and cell division.
d. Interphase has three phases: growth (G1), synthesis of DNA (S) and preparation for mitosis (G2).
e. During mitosis duplicated chromosomes line up in center with spindle fibers attached to help pull
them apart. Duplicated chromosomes are pulled apart by spindle fibers.
f. Cytokinesis-division of cytoplasm and reformation of cell membrane. Animal cell- pinches in
(cleavage) using microfilaments; plant cell- form cell plate reforms cell wall.
g. The cell cycle is directed by internal controls or checkpoints. Internal (enzymes and promoting
factors) and external signals (growth factors) provide stop and- go signs at the checkpoints. Ex.
Mitosis-promoting factor (MPF)
h. Cancer results from disruptions in cell cycle control (too much division, defective tumor suppressor
genes, overactive genes) which are a result of DNA damage to proto-oncogenes (regulatory genes)
which make products like cyclins and cyclin-dependent kinases.
i. Cells spend different amounts of time in interphase or division. Nondividing cells may exit the cell
cycle; or hold at a particular stage in the cell cycle.
j. Mitosis is used for growth and repair in animals; plants use mitosis to make gametes and for
growth or repair.
k. Mitosis usually begins with 1 cell, makes 2 identical cells or clones; maintains chromosome number;
1n1n or 2n2n.
l. Meiosis (occurs after interphase) takes diploid cells and reduces the chromosome number to
haploid. 2n1n.
m. During meiosis, homologous chromosomes are paired (one from mom and one from dad) and line
up in the center of the cell randomly. The homologues are pulled apart and separated in meiosis I.
A second division occurs in which the duplicated chromosomes are pulled apart.
n. Variation occurs in gametes during “crossing over,” and fertilization because of all possible
combinations of homologous chromosomes aligning during metaphase I.
2. Mendel’s Laws (remember he laid groundwork for genetics but these rules can all be broken looking at
chromosome theory and molecular genetics)
a. Law of Dominance- one allele will be expressed over another (ex. Aa – if big A is purple it will be
seen over little a which is white)
b. Law of Segregation- alleles pairs separate from each other during meiosis
c. Law of Independent Assortment- alleles assort independently during meiosis IF they are on
separate chromosomes (i.e. AaBb can make gametes AB, Ab, aB or ab)

Page 22 AP Biology: 2013 Exam Review
3. Probability, Patterns and Exceptions to Mendel’s Rules
a. product rule- multiply chance of one event happening by the chance of another event happening to
get the chance of both events occurring together
b. autosomal vs. sex-linked (on the X or Y chromosome)
c. monohybrid cross; one trait; 3:1 (Aa x Aa); 1:1 (Aa x aa) or 4:1 (AA x_), (aa x aa)
d. dihybrid cross; 9:3:3:1 genotype (AaBb x AaBb) or test cross 1:1:1:1(AaBb x aabb)
e. Thomas Hunt Morgan- fruit flies, X- linked traits
(1) male- heterozygous XY; Y chromosome is very small in mammals and fruit flies with few
genes
(2) female- homozygous XX
(3) single gene mutations on X chromosome cause disease such as hemophilia or
colorblindness
(4) sex limited traits are dependent on sex of individual like milk production or male patterned
baldness
f. incomplete dominance- red X white pink; both protein product are expressed and blended
g. codominance- red x white red and white; both protein products are equally expressed ex.AB
blood types
h. epistasis- one gene affects expression of another
i. linked genes- genes on same chromosome that are inherited together (can be unlinked by crossing
over); recombination frequency calculated by recombinants/total; used for chromosome mapping;
genes further apart cross over more often
j. gene/environment- phenotypes affect by environment, Siamese cat, flower color with soil pH,
seasonal color in arctic animals, human height and weight
k. polygenic- continuous variation, many genes affect one trait- height, color
4. Human Genetics
a. karyotype- 22 pair autosomes & 1 pair sex chromosomes + 46 total chromosomes
b. Chromosomal Mutations (occur during gamete formation)
(1) deletion, inversion, addition of genes as a result of crossing over mistakes
(2) chromosomal number abnormalities nondisjunction is failure of chromosomes to
separate at anaphase of meiosis
Vocabulary
anaphase
autosomal
cancer
cell cycle
cell division
centrioles
chromosome
codominance
crossing over
crossover frequency
cyclin-dependent kinase
cytokinesis
diploid (2N)
dominant
F1/F2 Generation
fertilization
gamete
genotype
haploid (1N)
heterozygous
homozygous
incomplete dominance
independent assortment
homologous chromosomes
independent assortment
interphase
meiosis
metaphase
mitosis
nuclear division
phenotype
prophase
recessive
recombination
segregation
sex chromosome
sex-linked
somatic cell
synapsis
synthesis
telophase

Page 23 AP Biology: 2013 Exam Review
Thinking Practice
1. Refer to the figure to the right.
a. What process is being shown in
this picture?
b. What type of organism are
these cells from? How do you
know?
c. Identify a numbered cell for
each of the four major stages of
mitosis.
d. In what stage are most of the cells in this image? What does this indicate about the amount of
time spent in each phase of the cell cycle?
2. Paclitaxel is a chemotherapy drug used to treat a variety of cancers. Paclitaxel inhibits both assembly and
disassembly of microtubules.
a. Which phase in the cell cycle is affected by Paclitaxel? How does this drug inhibit the growth of
cancer?
b. Paclitaxel affects not only cancer cells, but normal cells as well. Would the effects of Paclitaxel be
seen first in organs that have quickly dividing cells (like the intestine and hair follicles) or in organs
that have slow or nondividing cells (like muscles and the nervous system). Justify your reasoning.

Page 24 AP Biology: 2013 Exam Review
3. Two students debate about proteins
that regulate the cell cycle. One
argues that MPF triggers the
production of cyclin, while the other
argues that cyclin triggers the
production of MPF.
a. Based on the figure to the
right, which statement is
correct and why?
b. Propose a possible function of MPF, based on when it is produced in the cell cycle.
4. You have performed a dihybrid cross of plants and got the following data: 206 purple tall, 65 white tall, 83
purple short, 30 white short. Perform a chi-square analysis to test the null hypothesis that purple coloring
is dominant to white and tall height is dominant to short height.

Page 25 AP Biology: 2013 Exam Review
5. A space probe discovers a planet inhabited by creatures that reproduce with the same hereditary patterns
seen in humans. Three of the phenotypic characteristics of these creatures are: height, antennae, and nose
morphology. Earth scientists were able to do some controlled breeding experiments with these organisms.
100 males and 100 females were used in the experiments and the results of a number of crosses are
shown below. Review this information and use it answer the questions that follow.
Cross I: True-breeding (homozygous) tall creatures were crossed with true breeding short creatures. ALL of
the F1 were tall. The F1 creatures were crossed and the following data was obtained.
F2 Phenotype
Male
Female
Tall
2575
2625
Short
1425
1375
Cross II: True breeding creatures WITH antennae are crossed with true-breeding creatures WITHOUT
antennae. ALL of the F1 had antennae. The F1 creatures were crossed and the following data was
obtained.
F2 Phenotype
Male
Female
WITH antennae
3125
3100
WITHOUT antennae
875
900
Cross III: Creatures that are true breeding for upturned snout are crossed with creatures with down turned
snouts. ALL of the F1 offspring had upturned snouts. The F1 creatures were crossed and the following data
was obtained.
F2 Phenotype
Male
Female
Upturned Snout
1750
3475
Down Turned snout
1750
0
Cross IV: True breeding tall, with antennae creatures were crossed with true breeding short, without
antennae creatures. ALL of the F1 offspring were tall, with antennae. These F1 offspring were crossed with
true breeding short, without antennae creatures. The F2 data is in the table below.
F2 Phenotype
Male
Female
Tall, WITH antennae
2360
2220
Tall, WITHOUT antennae
220
300
Short, WITH antennae
260
220
Short, WITHOUT antennae
2240
2180
a. What conclusions can be drawn from cross I and II? Explain how the data supports your
conclusions (Hint! You might need to do a chi square analysis to support conclusions!)
b. What conclusions can be drawn from cross III? Explain how the data supports your conclusions
(Hint! You might need to do a chi square analysis to support conclusions!)
c. What conclusions can be drawn from cross IV? Explain how the data supports your conclusions
(Hint! You might need to do a chi square analysis to support conclusions!)

Page 26 AP Biology: 2013 Exam Review
Cell Cycle and Heredity Short Free Response (5 points)
Meiosis reduces chromosome number and rearranges genetic information. Explain how the reduction and
rearrangement are accomplished in meiosis.

Page 27 AP Biology: 2013 Exam Review
CONCEPT 5 – MOLECULAR GENETICS
1. DNA Structure
a. Discovery
(1) Avery-MacLeod- Marty- 1944 isolated DNA from Griffith’s transformation experiment
(2) Hershey-Chase- 1952 elegant experiment with virus and bacteria showing DNA was
injected not protein
(3) Watson, Crick, Wilkins, and Franklin- 1953 W and C published work showing structure of
DNA (used Wilkins and Franklins work to do so)
b. Structure of DNA
(1) Deoxyribose nucleic acid
(2) Double helix (two twisted stsrands) made of nucleotides (monomers)
(3) Nucleotide = phosphate + 5C deoxyribose sugar + nitrogen base
(4) Antiparallel strands- one runs 3’ to 5’ the other runs 5’ to 3’,sides of phosphates and
sugars (backbone), rungs of paired bases with hydrogen bonds in between
(5) Purines (adenine,guanine; double rings) pair with Pyrimidines (cytosine, uracil, thymine;
single ring)
(6) A - T- double H bond
(7) C – G- triple H bond
c. Location
(1) In eukaryotes DNA is found in nucleus on multiple linear chromosomes (a chromosome IS
a strand of DNA with proteins etc. associated).
(2) In prokaryotes DNA is not in a nucleus and is usually a single circular chromosome
(3) Prokaryotes, viruses, and eukaryotes (yeast) can contain plasmids (small extra-
chromosomal DNA that is double stranded DNA)
2. DNA replication
a. Process of making exact copies of DNA (i.e. for mitosis or meiosis)
b. Process is semi conservative (original strand is copied)
c. Steps
(1) Enzyme (helicase) unzip strands by breaking hydrogen bonds
(2) “Spare” nucleotides are added bidirectionally to bond complementarily with use of DNA
polymerases (DNA pol)
(3) DNA pol only can add to the 3’ to 5’ side and new DNA is made in the 5’ to 3’direction
(4) Replication bubbles open up and a replication fork is created because bubble is in half and it
has one side 3/5 and one 5/3
(5) RNA primers must be laid down to start process (RNA primase makes primers)
(6) Leading strand makes DNA continuously (3/5)
(7) Lagging strand makes DNA discontinuously (5/3), Okazaki fragments
(8) Lagging strand requires enzyme (ligase) to fuse fragments
3. RNA
a. Ribonucleic acid
b. Single stranded, different sugar called ribose, different base called uracil INSTEAD of thymine
c. Base pair rules in RNA, A-U and C-G
d. messenger RNA or mRNA carries information from DNA to the ribosome
e. transfer RNA or tRNA bind amino acids and are used in translation at ribosome

Page 28 AP Biology: 2013 Exam Review
f. ribosomal RNA or rRNA are part of ribosomes that have catalytic function
g. RNAi are molucules that are used for regulation of gene expression (turn on or off)
4. Transcription
a. making mRNA in nucleus
b. enzyme RNA pol reads the DNA in 3’ to 5’ direction and synthesizes complementary mRNA
c. Ex. 3’ to 5’ DNA is ATG CAT then the 5’ to 3’ mRNA made will be UAC GUA
d. Steps
(1) TATA Box where RNA pol binds and begins
(2) Transcription Factors (proteins that enhance transcription and help RNA pol into correct
shape)
(3) Elongation (adding of RNA nucleotides- does not stay attached to DNA)
(4) Termination, ends when RNA pol reaches a termination sequence
5. mRNA editing
a. introns are excised (cut out)
b. exons are left and spliced together using spliceosomes (snRNP’s)
c. add polyA tail to 3’
d. add GTP cap to 5’
e. each 3 are called a codon
f. go to ribosome (free or in RER)
6. Translation
a. mRNA code is read and matched with tRNA (brings amino acids) to construct a polypeptide using
the ribosome
b. Ex. mRNA codon is AAA then tRNA anticodon will be UUU and will have a corresponding amino acid
for that codon of mRNA
c. Initiation: 5’ end of mRNA attaches to small ribosome, tRNA with anticodon UAC attaches to start
codon AUG ; large ribosomal subunit binds and tRNA is in P site
d. Elongation: new tRNA enters A site; peptide bond forms when a.a. is transferred from tRNA in P
site to A site; translocation occurs and tRNA in A site moves to P
e. Termination: Ribosome encounters stop codon (UAA, UAG, UGA)
f. If in ER then: polypeptide is released into ER, then to Golgi complex, vesicle to cell membrane, then
exocytosis (may be given signals for exit/destination)
g. Free ribosomes typically make products for the cell and are not exported
7. Mutations
a. any change of DNA sequence, can be inheritable if it is in egg or sperm
b. point mutations- one nucleotide error; substitutions (i.e. A instead of G)
c. frame shift mutations- one or more bases deleted or inserted
d. silent mutations can occur, i.e. substitution codes for same a.a. or deletion/insertion is of three
nucleotides
Page 29 AP Biology: 2013 Exam Review
Vocabulary
amino acids
anticodon
base-pairing rules
cell differentiation
coding strand
codon
DNA
DNA ligase
DNA polymerase
DNA replication
exons
genetic code
helicase
hydrogen bonding
inducible genes
introns
lagging strand
leading strand
micro RNA (miRNA)
mutation
nucleic acids
nucleotides
Okazaki fragments
protein
replication fork
repressor
RNA (mRNA, rRNA, tRNA)
start codon/stop codon
template strand
transcription
transcription factors
translation
Thinking Questions
1. Compare the two DNA sequences shown below. Transcribe them into mRNA and translate them into an
amino acid sequence.
GTG CAC CTC ACT CCA GAG GAG (Normal Hemoglobin)
mRNA
amino acids
GTG CAC CTC ACT CCA GTG GAG (Sickle Cell Hemoglobin)
mRNA
amino acids
a. Circle any differences there are in the DNA, RNA and amino acid sequences that might exist between
these two sequences.
b. Identify the type of mutation that is represented AND EXPLAIN, IN DETAIL, what effect this would have
on the protein/pigment (be sure to mention the types of functional groups on the amino acids and
how this would affect shape of the molecule).
2. In prokaryotic cells, translation begins before transcription is finished. Give two reasons why this would
not be possible in eukaryotic cells.

Page 30 AP Biology: 2013 Exam Review
3. The restriction enzyme EcoRI cleaves double-stranded DNA at the sequence 5'-GAATTC-3' and the
restriction enzyme HindIII cleaves at 5'-AAGCTT-3'. A 20 kb circular plasmid is digested with each enzyme
individually and then in combination, and the resulting fragment sizes are determined by means of
electrophoresis. The results are as follows:
Make a diagram of the circular molecule and indicate the relative positions of the EcoRI and HindIII
restriction sites. (Hint: place one EcoRI site at '12 o'clock' and position the remainder relative to this site.)

Page 31 AP Biology: 2013 Exam Review
Molecular Genetics Short Free Response (4 points)
When DNA replicates, each strand of the original DNA molecule is used as a template for the synthesis of a
second, complementary strand. Compare and contrast the replication of the two new strands, listing and
explaining at least one similarity and one difference in the methods of synthesis. You may draw a diagram to
help answer the question, but be sure to explain your diagram in your answer.

Page 32 AP Biology: 2013 Exam Review
CONCEPT 6 – REGULATION
1. Feedback
a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable)
by regulating physiological processes, returning the changing condition back to its target set point.
b. Positive feedback mechanisms amplify responses and processes in biological organisms. The
condition initiating the response is moved farther away from the initial set-point. Amplification
occurs when the stimulus is further activated which, in turn, initiates an additional response that
produces system change.
2. Cell-to-cell communication
a. Cells receive or send inhibitory or stimulatory signals from other cells, organisms or the
environment.
b. In single-celled organisms it is response to its environment.
c. In multicellular organisms, signal transduction pathways coordinate the activities within individual
cells. Ex. Epinephrine stimulation of glycogen breakdown in mammals
d. Cells communicate by cell-to-cell contact. Ex Immune cells interact by cell-cell contact, antigen-
presenting cells (APCs), helper T-cells and killer T cells or plasmodesmata between plant cells that
allow material to be transported from cell to cell.
e. Cells communicate over short distances by using local regulators that target cells in the vicinity of
the emitting cell. Ex. Neurotransmitters, plant immune response
f. Signals released by one cell type can travel long distances to target cells of another cell type. Ex.
Hormones
g. A receptor protein recognizes signal molecules, causing the receptor protein’s shape to change,
which initiates transduction of the signal. Ex. G-protein linked receptors, ligand-gated ion channels,
tyrosine kinase receptors.
h. Signal transduction is the process by which a signal is converted to a cellular response. Signaling
cascades relay signals from receptors to cell targets, often amplifying the incoming signals, with the
result of appropriate responses by the cell.
i. Second messengers inside of cells are often essential to the function of the cascade.
j. Many signal transduction pathways include: Protein modifications or phosphorylation cascades in
which a series of protein kinases add a phosphate group to the next protein in the cascade
sequence.
3. Gene Regulation
a. Prokaryotes
(1) Inducers (turn genes on) and repressors (turn genes off) are small molecules that interact with
regulatory proteins and/or regulatory sequences.
(2) Regulatory proteins inhibit gene expression by binding to DNA and blocking transcription
(negative control).
(3) Regulatory proteins stimulate gene expression by binding to DNA and stimulating transcription
(positive control) or binding to repressors to inactivate repressor function.
b. Eukaryotes
(1) Transcription factors bind to DNA sequences and other regulatory proteins
(2) Some of these transcription factors are activators (increase expression), while others are
repressors (decrease expression).

Page 33 AP Biology: 2013 Exam Review
(3) The combination of transcription factors binding to the regulatory regions at any one time
determines how much, if any, of the gene product will be produced.
4. Immunity
a. Plants, invertebrates and vertebrates have multiple, nonspecific immune responses, ex: phagocytes
engulf and digest pathogens with the help of lysosomes
b. Mammals use specific immune responses triggered by natural or artificial agents that disrupt
dynamic homeostasis.
(1) The mammalian immune system includes two types of specific responses: cell mediated and
humoral.
(2) In the cell-mediated response, cytotoxic T cells, a type of lymphocytic white blood cell,
target‖intracellular pathogens when antigens are displayed on the outside of the cells.
(3) In the humoral response, B cells, a type of lymphocytic white blood cell, produce antibodies
against specific antigens.
(4) Antigens are recognized by antibodies to the antigen.
(5) Antibodies are proteins produced by B cells, and each antibody is specific to a particular
antigen.
(6) A second exposure to an antigen results in a more rapid and enhanced immune response.
5. Viruses
a. Replication
(1) Viruses inject DNA or RNA into host cell
(2) Viruses have highly efficient replicative capabilities that allow for rapid evolution
(3) Viruses replicate via the lytic cycle, allowing one virus to produce many progeny simultaneously
(4) Virus replication allows for mutations to occur through usual host pathways.
(5) RNA viruses lack replication error-checking mechanisms, and thus have higher rates of
mutation.
(6) Related viruses can combine/recombine information if they infect the same host cell.
(7) Some viruses are able to integrate into the host DNA and establish a latent (lysogenic) infection
(8) HIV is a well-studied system where the rapid evolution of a virus within the host contributes to
the pathogenicity of viral infection.
(9) Genetic information in retroviruses is a special case and has an alternate flow of information:
from RNA to DNA, made possible by reverse transcriptase, an enzyme that copies the viral RNA
genome into DNA. This DNA integrates into the host genome and becomes transcribed and
translated for the assembly of new viral progeny.
Vocabulary
Antibody
Antigen
B-cell
Cell-mediated immunity
communication
cyclic AMP (cAMP)
cytotoxic T-cell
G-protein linked receptor
Helper T-cell
Hormone
Humoral immunity
Inducer
Lytic cycle
Lysogenic cycle
Negative feedback
Operon
Operator
Phagocyte
phagocytosis
phosphorylation cascade
positive feedback
protein kinase
receptor
repressor

Page 34 AP Biology: 2013 Exam Review
retrovirus
reverse transcriptase
second messenger
signal cascade
signal transduction
signal transduction pathway
transcription factor
virus
white blood cell
Thinking Practice
1. Refer to the diagram at the right to
respond to the following questions.
a. Is the hormone hydrophobic or
hydrophilic? How do you know?
b. Explain how the action of the
hormone might be different if it
could move through the cell
membrane.
c. Explain what is happening in this picture and make a prediction about what will be the end result in
the cell to which this hormone has bound.
2. Lactose digestion in E. coli begins with its hydrolysis by the enzyme b-galactosidase. The gene encoding b-
galactosidase, lacZ, is part of a coordinately regulated operon containing other genes required for lactose
utilization. Use the legend below to draw the gene and its interaction with RNA polymerase, the repressor
protein, and lactose when lactose is being digested.

Page 35 AP Biology: 2013 Exam Review
3.
Based on the information provided, propose a possible mechanism for a drug to resist HIV infection.
4. Describe the processes occurring at each of the
numbered positions (I, II, III, and IV) in the diagram
to the right.

Page 36 AP Biology: 2013 Exam Review
5. Refer to the images at the right to answer the following:
a. Which immune response in shown: cell
mediated or humoral? Explain how you
know.
b. What are the “Y” shaped molecules
called? What is their role in the immune
response?
c. Describe how the “Y” shaped molecules relate
to the graph displayed.
6. One student described an action potential in a neuron by saying “As more gates open the concentration of
sodium inside the cell increases and this causes even more gates to open.” Is this an example of a positive
or negative feedback loop? Justify your reasoning.
7. The figure to the right shows the feedback mechanism for regulating
blood glucose.
a. Is this a positive or negative feedback loop? Explain your
answer.
b. Individuals that suffer from Type I diabetes do not have
functional insulin-producing cells. Describe how their blood
will differ from that of a healthy individual after a glucose-rich
meal.

Page 37 AP Biology: 2013 Exam Review
8. In a molecular biology laboratory, a student obtained competent E. coli cells and used a common
transformation procedure to induce the uptake of plasmid DNA with a gene for resistance to the antibiotic
kanamycin. The results below were obtained.
a. What is the purpose of Plate IV?
b. Explain the growth you see and the type of bacteria (transformed vs. non-transformed or both)
that would be on Plate 1.
c. Explain the growth you see and the type of bacteria (transformed vs. non-transformed or both)
that would be on Plate II.
d. If the student repeated the experiment, but the heat shock was unsuccessful and the plasmid was
unable to be transformed, for which plates would growth be expected? Explain your answer.

Page 38 AP Biology: 2013 Exam Review
CONCEPT 7 – EVOLUTION
1. Natural Selection
a. Major mechanism of change over time – Darwin’s theory of evolution
b. There is variation among phenotypes – genetic mutations play a role in increasing variation
c. Competition for resources results in differential survival, with individuals with the most favorable
traits surviving to reproduce offspring
d. An adaptation is a genetic variation that is favored by selection and is manifested as a trait that
provides an advantage to an organism in a particular environment.
e. Fitness is the ability to survive and reproduce
2. Hardy-Weinberg Equilibrium
a. A mathematical model used to calculate changes in allele frequency, providing evidence for the
occurrence of evolution in a population.
b. 5 conditions must be met for a population to be in HW equilibrium – conditions are seldom met
(1) Large population
(2) No migration
(3) No mutations
(4) Random mating
(5) No natural selection
c. Equations
(1) p = the frequency of dominant alleles in a population
(2) q = the frequency of recessive alleles in a population
(3) p
2
= the frequency of homozygous dominant individuals in a population
(4) q
2
= the frequency of homozygous recessive individuals in a population
(5) 2pq=the frequency of heterozygous individuals in a population
(6) p + q = 1
(7) p
2
+ 2pq + q
2
= 1
3. Speciation
a. An evolutionary process by which 2 or more species arise from 1 species and 2 new species can no
longer breed and reproduce successfully
b. Many mechanisms by which it can occur
(1) Geographic isolation
Species separated by physical barrier
(2) Reproductive isolation
Different behaviors limit mating
Different habitats limit mating
Different mating seasons limit mating
Different anatomical structures limit mating
c. Can take place over millions of years or rapidly (after extinction events, for example)
4. Evidence for Evolution
a. Fossils can be dated by a variety of methods that provide evidence for evolution. These include the
age of the rocks where a fossil is found, the rate of decay of isotopes including carbon-14, the
relationships within phylogenetic trees, and the mathematical calculations that take into account
information from chemical properties and/or geographical data.

Page 39 AP Biology: 2013 Exam Review
b. Morphological homologies represent features shared by common ancestry. Vestigial structures are
remnants of functional structures, which can be compared to fossils and provide evidence for
evolution.
c. Biochemical and genetic similarities, in particular DNA nucleotide and protein sequences, provide
evidence for evolution and ancestry.
5. Origin of Life
a. Primitive Earth provided inorganic precursors from which organic molecules could have been
synthesized due to the presence of available free energy and the absence of a significant quantity
of oxygen.
b. Chemical experiments have shown that it is possible to form complex organic molecules from
inorganic molecules in the absence of life.
c. These complex reaction sets could have occurred in solution (organic soup model) or as reactions
on solid reactive surfaces.
d. The RNA World hypothesis proposes that RNA could have been the earliest genetic material.
6. Phylogenetic Trees
a. Phylogenetic trees and cladograms can represent traits that are either derived or lost due to
evolution.
b. Phylogenetic trees and cladograms illustrate speciation that has occurred, in that relatedness of
any two groups on the tree is shown by how recently two groups had a common ancestor.
c. Phylogenetic trees and cladograms can be constructed from morphological similarities of living or
fossil species, and from DNA and protein sequence similarities.
d. Phylogenetic trees and cladograms are dynamic, constantly changing due to current and emerging
knowledge.
Vocabulary
Adaptation
Bottleneck Effect
Common Ancestor
Cladogram
Evolution
Gene Flow
Gene Pool
Genetic Drift
Geographic Isolation
Fitness
Hardy-Weinberg Equilibrium
Natural Selection
Morphology
Phylogenetic Tree
Reproductive Isolation
Speciation
Variation
Page 40 AP Biology: 2013 Exam Review
Thinking Questions
1. As a field researcher you are sent to the Arizona desert to study the prairie dog species C. ludivincianus to
determine if the population is in Hardy-Weinberg equilibrium. Specifically, you are studying this population
with respect to the gene that determines the coat color in C. ludivincianus. This trait is coded for by a
single gene (the NDY6 gene) with two alleles (N, n) and is passed down from one generation to the next.
After sampling 170 of these prairie dogs, you find that 36% of the C. ludivincianus population is
homozygous recessive for coat color. Assuming that the population is in Hardy-Weinberg equilibrium…
a. What is the allele frequency of the N allele?
b. What is the frequency of homozygous dominant prairie dogs?
c. What is the frequency of heterozygous prairie dogs?
d. What conditions must be being satisfied?
2. Sixty flowering plants are planted in a flowerbed. Forty of the plants are red-flowering homozygous
dominant. Twenty of the plants are white-flowering homozygous recessive. The plants naturally pollinate
and reseed themselves for several years. In a subsequent year, 178 red-flowered plants, 190 pink-flowered
plants, and 52 white-flowered plants are found in the flowerbed. Use a chi-square analysis to determine if
the population is in Hardy-Weinberg equilibrium.

Page 41 AP Biology: 2013 Exam Review
3. For the past 10 to 25 years, farmers have planted crop seeds that have been genetically modified to
withstand treatment with a common weed killer called Roundup®. This allows the farmers to spray their
fields to get rid of weeds without harming their crops. Recently, more and more farmers have discovered
that their fields have Roundup-resistant pigweed growing along with their crop. Describe what has most
likely happened over time to lead to this.
4. Peppered moths have wings that vary in color, ranging from white to dark gray. During the Industrial
Revolution through the mid-20th century, factories and power plants, which burned coal, produced large
quantities of soot and smog. Near industrialized areas, black powder covered surfaces, including the moth
habitat.
a. Use this information to explain the changes seen in light and dark peppered moths from 1800-
1950, as shown in the graph below.
b. Propose an explanation for the return of the peppered moth population to more light than dark
moths by the year 2000.
Page 42 AP Biology: 2013 Exam Review
5. Five new species of bacteria were discovered in Antarctic ice core samples. The nucleotide (base) sequences
of rRNA subunits were determined for the new species. The table below shows the number of nucleotide
differences between the species. Draw a phylogenetic tree indicating the relatedness of these 5 species.
Species
1
2
3
4
5
1
-
2
23
19
17
2
-
24
19
18
3
-
23
23
4
-
1
5
-
6.
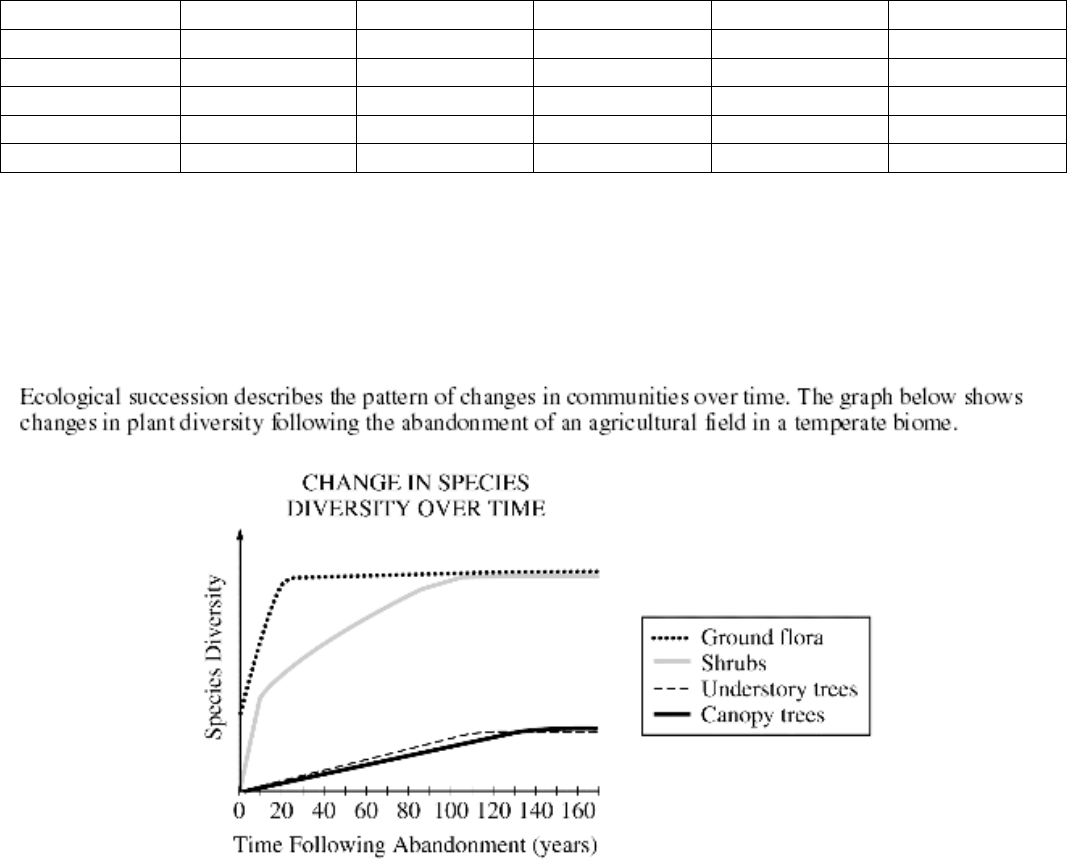
a. Discuss the differences in diversity among the plants shown in the graph.
b. If a scientist wanted to determine if two understory trees in the field were the same species, what
pieces of evidence would she gather and how would these inform her conclusion?

Page 43 AP Biology: 2013 Exam Review
Evolution Long Free Response (10 points)

Page 44 AP Biology: 2013 Exam Review

Page 45 AP Biology: 2013 Exam Review

Page 46 AP Biology: 2013 Exam Review
CONCEPT 8 – ECOLOGY
1. Populations
a. group of individuals of same species living in same area (size, density, distribution/dispersion)
b. habitat (type of area organism lives) vs. niche (role in ecosystem)
c. competition for resources
d. age structure (rapid growth vs. declining vs. stable populations)
e. population growth
(1) density dependent limiting factors (competition for resources, parasites & diseases, waste
products, stress, predation)
(2) density independent limiting factors (climate = temperature & rainfall, natural disaster)
(3) exponential growth (J-shaped, unlimited) vs. logistic growth curve (S-shaped, limited)
(4) carrying capacity = maximum population supported by habitat
(5) populations can cycle
f. Population ability to respond to changes in the environment is affected by genetic diversity. Species
and populations with little genetic diversity are at risk for extinction.
2. Communities
a. measured and described in terms of species composition and species diversity
b. symbiosis = species interaction
(1) mutualism +/+ (acacia tree & ants; lichens, N-fixing bacteria & legume plants)
(2) commensalism +/0 (egrets & cattle)
(3) parasitism +/– (tapeworm, cowbird)
(4) predation +/– (carnivores & herbivores)
(5) competition
3. Ecosystems
a. Free Energy
(1) Reproduction and rearing of offspring require free energy beyond that used for maintenance
and growth. Different organisms use various reproductive strategies in response to energy
availability.
(2) There is a relationship between metabolic rate per unit body mass and the size of multicellular
organisms — generally, the smaller the organism, the higher the metabolic rate.
(3) Excess acquired free energy versus required free energy expenditure results in energy storage
or growth.
(4) Insufficient acquired free energy versus required free energy expenditure results in loss of mass
and, ultimately, the death of an organism.
b. Energy flow/production = energy flows through; 90% lost at each level & 10% transferred to next
level
(1) trophic levels = primary producers, primary consumers, secondary consumers, tertiary
consumers, detritivores & decomposer
(2) ecological pyramids (pyramids of energy, biomass, numbers)
(3) food chains & food webs
(4) Biotic and Abiotic factors can both cause disruption and collapse of ecosystems

Page 47 AP Biology: 2013 Exam Review
Vocabulary
Apex predator
Biomagnification
Biomass
Carrying capacity
Carnivore
Commensalism
Competition
Consumer
Density-dependent
Density-independent
Decomposer
Detrivore
Energy pyramid
Exponential growth
Food chain
Food web
Herbivore
Heterotroph
Logistic growth
Mutualism
Trophic level
Parasitism
Population
Predation
Primary productivity
Producer
Thinking Questions
1. Invasive species are species that are introduced into an environment but are not naturally found in that
environment. One example of an invasive species is the American gray squirrel, introduced into Britain at
the end of the 18th century. Until 1876 the only native squirrel in Britain was the European red squirrel,
which was found in deciduous and coniferous forests. By 1940 the gray squirrel had displaced the red
squirrel across most of the British Isles, and by 1984 the red squirrel was only found in isolated coniferous
woodland areas. After its initial introduction, the gray squirrel population increased rapidly; however, in
recent years population sizes within specific environments have become stable.
a. Explain why the newly-introduced gray squirrel initially showed rapid population growth and why
the native red squirrel showed a population decline.
b. Why has the population size of the gray squirrel become stable in recent years?
2. The first age structure graph below for country X shows the percent of the population in each age group
for the year 2000. The remaining three graphs are projections of how the age structure of country X will
change. From these age structure diagrams construct a graph of population size vs. time for 2000-2080
and justify your prediction.

Page 48 AP Biology: 2013 Exam Review
3. The graphs below display the growth rate for two species of bacteria when grown separately and together.
a. The population growth of which bacteria is more affected by growing conditions? Explain how you
know.
b. Using the information provided in the graphs, make a prediction as to why the bacteria identified in
part a is more affected by growing conditions than the other bacteria.
4. Interdependence in nature is illustrated by the transfer of energy through trophic levels. The diagram
below depicts the transfer of energy in a food web of an Arctic lake located in Alaska.
a. Identify an organism from each of the 5 trophic levels (producer, primary consumer, secondary
consumer, teritiary consumer and decomposer) and explain how energy is obtained at each level.
b. Describe the efficiency of energy transfer between trophic levels of this food web.
Page 49 AP Biology: 2013 Exam Review
c. Explain how the amount of energy available at each trophic level affects the size of each population.
d. If the cells in the dead terrestrial plant material that washed into the lake contained a commercially
produced toxin, what would be the likely effects of this toxin on this food web? Explain.
e. If all of the Sculpin in this ecosystem were removed, predict how it would impact the following and
explain each prediction:
The population of lake trout
The population of snails
The population of algae
The amount of oxygen produced in the ecosystem
The amount of light energy absorbed by the ecosystem

Page 50 AP Biology: 2013 Exam Review
Ecology Long Free Response (10 points)

Page 51 AP Biology: 2013 Exam Review

Page 52 AP Biology: 2013 Exam Review

Page 53 AP Biology: 2013 Exam Review
FREE RESPONSE GRADING RUBRICS
Biochemistry Short Free Response – Page 5
Cell Long Free Response – Pages 10-12
Part A
1 point correct orientation with dependent variable (concentration) on y (vertical) axis and
independent variable (time) on x (horizontal) axis
1 point correct axes labels with units and scaling for 5% line on axes provided
1 point correct plotting of all data points including zero (0,0); line is not necessary but if drawn must
not extend beyond last data point; dashing line beyond last data point is okay; arrow at end of
line is okay.
Part B
1 point correct prediction and legend (or label) for 0%, 1%, and 10% lines (0% line flat, 1% line below
5% line, 10% line above 5% line)
1 point correct explanation for 0% line (e.g., since there is no NaCl in the bag no Na+Cl- can diffuse into
the water in the beaker)
1 point correct explanation for 1% line — must include a discussion of rate; connects concentration of
NaCl with diffusion rate
1 point correct explanation for 10% line — must include a discussion of rate; connects concentration of
NaCl with diffusion rate
Part C
1 point statement that water will leave the plant and description of effect this has on plant cell (e.g.,
loss of turgor, plasmolysis, decrease in cell volume, decrease in central vacuole volume)

Page 54 AP Biology: 2013 Exam Review
1 point concept of osmosis (e.g., movement of water across a selectively permeable membrane (cell or
cell membrane) from solution with lower solute concentration (hypotonic) to solution with
higher solute concentration (hypertonic)
1 point explanation that water moves from solution with higher (more positive/less negative) water
potential (ψ) to solution with lower (more negative) water potential (ψ)
1 point explanation of how water loss can cause decreased crop production (e.g., stomates close,
transpiration stops, photosynthesis stops, decreased metabolism)
Energy and Metabolism Short Free Response – Page 19
Cell Cycle and Heredity Short Free Response – Page 25

Page 55 AP Biology: 2013 Exam Review
Molecular Genetics Short Free Response – Page 30
SIMILARITIES
2 POINTS MAXIMUM
1 point for similarity, 1
point for elaboration
(in parentheses)
• Synthesize DNA in 5’ to 3’ direction (DNA polymerase III can only work in one
direction)
• Use RNA primers to initiate replication (primase, DNA polymerase III must
have a started sequence to be functional)
• Both have RNA primers replaced with DNA (DNA polymerase I)
DIFFERENCES
2 POINTS MAXIMUM
2 points for difference
including description of
both strands
• One strand is synthesized as one, continuous strand (leading strand), while
the other is synthesized in fragments (lagging strand).
• Lagging strand must use ligase to connect segments of DNA, while leading
strand does not require use of this enzyme.
Evolution Long Free Response – Pages 42-44

Page 56 AP Biology: 2013 Exam Review
Ecology Long Free Response – Pages 49-51
